Nasim Afsar
Brian Anderson
Wanda Barfield
Monica Bharel
Jeffrey Brown
Peter Embí
Adam Eschenlauer
William Gordon
Susan Gregurick
Brent James
Anupam Jena
Peter Lee
Thomas Maddox
Kenneth Mandl
Ravi Parikh
Larra Petersen-Lukenda
Troy Sarich
Abdul Shaikh
Peter Speyer
Kenneth Yale
Commission on Investment Imperatives for a Healthy Nation
Executive Summary
Digital data streams interface seamlessly across much of today’s interconnected society, powering advancements across global communications, financial transactions, and retail sales. However, despite near-universal adoption of electronic health records (EHRs) and technological advances and regulatory impetus needed to spur health data interoperability, the health sector has not realized the full potential of data digitization. The authors of this discussion paper have found that the absence of a cohesive digital and data architecture is preventing the full realization of interoperability’s benefits, slowing innovation, impeding patient-centered care, and enabling digital health infrastructure’s persistent fragmentation.
Robust interoperability is necessary but not sufficient. The authors of this discussion paper have found that progress in achieving interoperability now requires moving from a sole focus on data exchange to a more intentional digital and data architecture that establishes a common language and set of protocols for multimodal technology and data use; defines boundaries for modularity in systems, markets, and regulation; gives purpose to interoperability standards; and enables key cross-cutting features such as data liquidity and user-centered design. To assist the reader in better understanding these issues, this paper includes detailed comparisons with other highly regulated industries that have undergone digital transformation and achieved success in one or more of these cross-cutting areas. The authors have also included specific case studies of how artificial intelligence (AI)-driven predictive analytics, telehealth, remote patient monitoring, and data sharing could create opportunities for better care coordination, reduced costs, and effective discovery for health concerns including cardiovascular disease, maternal and fetal health, cancer care, and diabetes. The authors also recognize the importance of aligning health care payment incentives with interoperable infrastructure and have emphasized a deregulatory approach that prioritizes interoperability standards over prescriptive EHR functionality.
In recent years, there has been significant progress in the development of data and technology standards. The authors thus urge “moving up the stack” to address known drivers of system misalignment, with particular focus on four areas:
| Misalignment Driver | Levers to Achieve Alignment and Drive Progress |
|---|---|
| Regulatory Complexity |
|
| Industry Fragmentation |
|
| Misaligned Financial Incentives |
|
| Resistance to Innovation |
|
Table 1 | Levers to Achieve Alignment and Drive Progress for Digital and Data Architecture
SOURCE: Created by authors.
Introduction
Digital data undergirds many facets of today’s interconnected society, including e-commerce, instant messaging, social media, and virtual banking (Boston Consulting Group, 2023). Digital health—defined as computing platforms, connectivity, software, and sensors for health care and related uses—has permeated nearly every aspect of health care, including primary care, consumer health, public health surveillance, health education, specialty care, long-term care, behavioral health, pharmaceuticals, medical devices, and value-based care arrangements (U.S. Food & Drug Administration, 2020). However, the health sector continues to lag in developing the robust digital health infrastructure necessary to fully realize these innovations, limiting potential gains in efficiency, access, prevention, diagnosis, treatment, discovery, and public health outcomes.
The digital health infrastructure is underdeveloped despite the health care industry generating 30 percent of the world’s data volume, equating to 2.3 zettabytes—one trillion gigabites—of data annually (Moore and Guichot, 2024). Despite the rapid production of newer and better medical evidence, the health system is currently unable to fully harness this knowledge for high-quality care. A typical hospital produces 50 petabytes—50 million gigabites—of data per year, including clinical notes, lab tests, medical images, sensor readings, genomics information, and operational and financial data (World Economic Forum, 2019). However, only 3 percent of these data are used for AI, analytics, downstream applications, and coordination across the health ecosystem that could improve patient experience, produce efficiencies, and result in better health outcomes (Moore and Guichot, 2024).
Currently, hospitals, public health organizations, policymakers, regulators, and other health facilities rarely have the capability to utilize health-relevant data collected outside clinical settings—including patient-reported information and data from wearables like smartwatches and rings (McGraw and Mandl, 2021). Furthermore, there are currently few mechanisms to collect insights into the social determinants of health—such as transportation access, education, and income—and environmental determinants like air pollution, food and shelter access, surface temperatures, chemical hazards, and animal and vector exposures.
High-quality digital and data solutions can have significant positive impacts on health and health care. In clinical settings, timely access to accurate and complete clinical data integrated with real-time CDS has been shown to improve care delivery performance, enhancing both timeliness and patient experience (Poissant et al., 2005). At a population health level, high-quality, real-time, interoperable data is essential for effective public health management, including responses to pandemics and crises such as the opioid epidemic (Galea and Abba-Aji, 2024). The substantial influence of the social determinants of health underscores the need for holistic data collection to inform strategies that support healthier behaviors, reduce the burden of chronic diseases, and empower individuals and communities to achieve better health outcomes.
Digital health interventions have demonstrated wide-ranging favorable effects on health care spending across a range of areas, including video monitoring service systems, text messaging interventions, web platforms, digital health portals, telephone support, and mobile phone-based systems and applications (Gentili et al., 2022). Perhaps most notably, most current commercial EHR systems were originally built to replace a hand-written medical record and maximize financial performance in a fee-for-service payment environment (Sinsky et al., 2020). Clinical operations, process execution, changes and improvements in patient quality, automating data entry and calculation to reduce workforce burdens, and identifying errors to prevent future problems were added as secondary and tertiary functions within that initial financially-focused framework.
The lack of an overarching digital and data architecture that enables the use of multimodal clinical and biological data alongside information about the social and environmental drivers of health is a fundamental obstacle to realizing the full benefit of digital health tools. This gap also results in a health care technology industry that is less well-organized and productively innovative than other regulated industries that have undergone digital transformation. Additionally, the legal and regulatory framework for current and future EHR development is woefully inadequate and trails behind technology developments. An optimal digital and architecture requires an organized and mature framework that allows for future innovation. If left unaddressed, this lack of digital and data architecture will stifle medicine’s progress, reduce health care efficiency, increase the potential for automation bias, disrupt the health workforce ecosystem, limit quality improvement and medical error reduction, and widen gaps in care for low income and rural settings (Zhao et al., 2024).
The underlying framework for this discussion paper is the Learning Health System’s Shared Commitments Trust Framework (NAM, 2025c). The Framework has been stewarded by the National Academy of Medicine (NAM) since 2006 when the concept of a LHS was conceived and embedded in the charter of what is now the NAM Leadership Consortium (NAM, 2025b). A LHS is defined as one in which science, informatics, incentives, and culture are aligned for continuous improvement, innovation, and equity—with best practices and discovery seamlessly embedded in the delivery process, individuals and families as active participants in all elements, and new knowledge generated as an integral by-product of the delivery experience (McGinnis et al., 2024).
Table 2 presents the Shared Commitments, which provide common ground aims for every organization engaged in the advancement of health.
| Engaged | Gives primacy to understanding, caring, and acting on people’s goals. |
| Safe | Deploys verified protocols to safeguard against risk from unintended harm. |
| Effective | Applies continuously updated evidence to target goal achievement. |
| Efficient | Delivers optimal outcomes and affordability for accessible resources. |
| Fair | Advances parity in individual opportunity to reach full health potential. |
| Accessible | Provides timely, convenient, interoperable, and affordable services. |
| Accountable | Identifies clear responsibilities, measures that matter, and reliable feedback. |
| Transparent | Displays full clarity and sharing in activities, processes, results, and reports. |
| Secure | Embeds safeguards in access, sharing, and use of data and digital/AI tools. |
| Adaptive | Centers continuous learning and improvement in organizational practices. |
Table 2 | Shared Commitments: A Trust Framework for Continuously Learning Health System
SOURCE: NAM. n.d. Learning health system shared commitments. Available at: https://nam.edu/our-work/programs/leadership-consortium/learning-health-system/ (accessed January 14, 2026).
To help inform a path forward, including the work of the Commission on Investment Imperatives for a Healthy Nation, the National Academy of Medicine (NAM) convened an expert working group to outline a shared vision and potential actions for improving industry fragmentation around digital infrastructure and ensuring that the potential of data digitization is realized. As part of this core effort to advance a LHS and foster alignment within the health sector toward a value-based health care system, the author group:
- Examined the current landscape of data and digital health in the health care sector,
- Highlighted knowledge gaps in need of further exploration,
- Identified the health and economic costs of maintaining the status quo,
- Identified the legal, regulatory, cultural, and other levers available to increase alignment, and
- Outlined strategies to build the will to apply the levers to the right opportunities.
Digital Health Landscape
The US health data infrastructure is a complex system that attempts to support health care data interoperability, quality measurement and improvement, use in public health, privacy and security, and health care access. However, it suffers from the lack of a comprehensive digital and data architecture that can unify and organize the continual technological evolution of the disparate systems and standards used across the ecosystem (Abernethy et al., 2022). This gap hampers the efficient flow of information, leading to fragmented data silos. Such fragmentation not only reduces technical capability but also hampers innovators in deploying new technologies system-wide. An overarching and flexible digital and data architecture could establish a common language and set of protocols for all systems, enabling seamless data exchange and integration.
This section describes how health care’s current data infrastructure impedes interoperability, quality measurement and improvement, highlighting the current state of and future directions for improving the health data landscape.
Interoperability
Interoperability refers to the ability of different health information systems, devices, and applications to access, exchange, integrate, and cooperatively use data in a coordinated manner within and across organizational boundaries, providing timely and seamless portability of information (HIMSS, n.d.). It is a critical component for achieving a fully integrated health system.
Rates of EHR and other digital tool adoption have been rising, driven by incentives from the Health Information Technology for Economic and Clinical Health (HITECH) Act and the Meaningful Use program (Blumenthal, 2011). However, adoption rates vary widely among health care providers, with larger health systems and hospitals showing higher rates compared to smaller practices (Charles et al., 2014). Interoperability challenges arise from the wide range of health data—spanning biomedical, clinical, community, and public health organizations—and the varied priorities of health system stakeholders. Differences in data formats, terminologies, and standards across EHRs, laboratory systems, imaging systems, payment systems, quality measures, wearables, and other digital health information systems impede seamless data exchange. Additionally, health care providers, payers, patients, caregivers, and regulatory bodies often have conflicting goals and requirements for managing patient privacy and achieving objectives beyond health care delivery—including clinical trial matching, disease surveillance, post-market monitoring of regulated products, quality and value measures, discovery research, or nudging lifestyle adjustments to improve well-being and prevent disease—that prevent cohesive movement toward a singular goal (Adler-Milstein and Jha, 2014).
Ensuring patient access to their health data and secure data sharing among providers has been a central priority for ONC and, more recently, the ASTP (see Figure 1). These efforts aim to empower patients, enhance care quality, and support streamlined, secure health information exchange across sectors. While significant interoperability exists today, with nationwide networks conducting 40 to 50 million daily transactions of standardized medical records, critical gaps remain in achieving the seamless, computable data exchange needed for a true LHS (ONC/ASTP, 2023).’
Figure 1 | ASTP Foundational Elements for Interoperable Health Data Exchange
SOURCE: Image created by authors.
Regulatory Foundations for Interoperability
A central focus of the ASTP/ONC has been implementing provisions of the 21st Century Cures Act, which requires that patients can use certified health information technology to access all elements of their health records via an API without “special effort” (Mandl and Kohane, 2017). ASTP/ONC’s pivotal rulemaking includes the 21st Century Cures Act Rule, which specifies the “how” in removing barriers to interoperability and expanding the scope of data sharing (21st Century Cures Act, 2020; Mandl and Kohane, 2020). The Rule combats information blocking to ensure that patients and providers have seamless access to health data and establishes a federal framework for secure and interoperable exchange.
Core FHIR APIs for Interoperability
A small set of standardized APIs—SMART on FHIR for individual-level access, Bulk FHIR for population-level exchange, and the EHI Export capability for full-record portability—form the essential technical backbone of a modern digital health ecosystem (Mandel et al., 2016). Together, they enable consistent and computable data flow across clinical, administrative, research, and patient-facing applications. As part of this backbone, the United States Core Data for Interoperability (USCDI) defines the nationally-required set of clinical data classes and elements that certified FHIR APIs must support—including the full text of patient clinical notes, a critical component in the era of natural language processing and large language models (ASTP, 2025a).
CMS’s Interoperability and Prior Authorization Final Rule extends API-based interoperability beyond EHRs by requiring Medicare Advantage, Medicaid, Children’s Health Insurance Program (CHIP), and Exchange plans to implement FHIR R4 APIs for prior authorization, payer-to-payer exchange, and provider access effective January 1, 2027 (Centers for Medicare & Medicaid Services, 2024). These requirements apply to public payers as well as private insurers operating Medicare Advantage, Medicaid managed care, CHIP managed care, and Exchange plans, making it the first federal rule to require FHIR-based interoperability within the private insurance market. The Rule bridges clinical and claims data exchange and advances a shared digital and data architecture across provider EHRs and CMS’s value-based care initiatives. It also builds on CMS’s earlier Interoperability and Patient Access Final Rule, which established the first FHIR-based Patient Access API and Provider Directory API requirements for Medicare Advantage, Medicaid managed care, and Exchange plans, forming the foundation for CMS’s expanding FHIR ecosystem (Centers for Medicare & Medicaid Services, 2020).
SMART on FHIR: Individual-Level Access and App Integration
SMART on FHIR provides a uniform mechanism for patients and clinicians to connect applications to EHRs (Mandel et al., 2016). By making standardized data transparent—including structured elements and clinical notes—the program supports patient-mediated exchange, consumer applications embedded into major mobile operating systems, and clinician-facing applications that run directly within EHR workflows (Mandl and Kohane, 2009; Mandl and Kohane, 2016). The value of this mechanism lies not in the mechanics of the protocol but in enabling a flexible ecosystem where tools can work across health systems without custom integration.
Bulk FHIR: Scalable Population-Level Data Exchange
Bulk FHIR enables standardized extraction of population-level clinical data without the custom mapping traditionally required for research, public health, or value-based care initiatives (Mandl et al., 2020; Boston Children’s Hospital, 2022; McMurry et al., 2024). Early deployments of this mechanism have demonstrated the potential for “push-button population health,” but have also unearthed performance constraints that underscore the need for continued maturation to ensure speed, scalability, and timeliness comparable to traditional methods of exporting data (Jones et al., 2024). Toolkits have emerged to assess data quality and performance, supporting more consistent use of the API across diverse health systems (EHR Good Neighbor, n.d.).
However, Bulk FHIR remains a developing capability. Few EHR systems use FHIR as their native storage model and the standard currently lacks a fully unified approach for defining and exchanging patient cohorts. These issues create operational and architectural challenges when handling large, ad-hoc population-level extracts. Yet, FHIR’s value lies precisely in providing a common, well-documented interoperability layer across otherwise incompatible systems—an essential alternative to today’s institution-specific proprietary exports that require repeated, site-by-site data mapping. Early implementations of the mechanism also show that high-performance Bulk FHIR export is achievable. For example, instances such as Regenstrief Institute’s implementation demonstrate rapid export rates when appropriately engineered, and several organizations have reported that running exports against analytic servers rather than transactional systems can fully isolate performance impact (McMurry et al., 2024). While underinvestment has resulted in uneven performance across current EHR implementations, work focused on new standards for cohort-definition APIs and the growing use of Bulk FHIR in production environments underscore both the feasibility and the momentum toward widespread adoption of this approach. Continued regulatory and market pressure can help ensure that vendors invest in scalable and reliable Bulk FHIR capabilities that serve patients, providers, and the broader LHS.
EHI Export: Full-Record Portability and Completeness
The 21st Century Cures Act’s requirement for EHI Export capability aims to help ensure comprehensive access to an individual’s entire health record—including unstructured content, non-USCDI elements, and historical data—via standardized mechanisms aligned with patient privacy and access rights. However, the current certification criterion requires vendors to make full record exports available but does not specify an API for patient-initiated, application-based retrieval. This gap limits the ease with which individuals and their applications can obtain a complete copy of their records (Phelan et al., 2024). Full-record portability remains especially important for AI-supported care and transitions requiring longitudinal clinical detail.
Complementary Standards That Enhance Practical Use
Workflow-oriented standards such as CDS Hooks and FHIR subscriptions extend the functionality of SMART and Bulk FHIR by enabling event-driven alerts, CDS calls, and real-time notifications (HL7 International, 2022; Assistant Secretary for Technology Policy, 2025). These complementary standards enhance usability and timeliness, enabling information to surface at the right moment in clinical workflow.
Trusted Exchange Framework and Common Agreement (TEFCA) – A Nationwide Exchange
Authorized by the 21st Century Cures Act and formalized through the Health Data, Technology, and Interoperability Final Rule, TEFCA establishes a nationwide “network-of-networks” to enable query-based exchange across health systems, payers, and public health agencies (Assistant Secretary for Technology Policy, 2025). TEFCA builds on and connects long-standing exchange networks such as Epic Care Everywhere, Carequality, and CommonWell, offering a unified set of policies and “rules of the road” to enable more consistent nationwide exchange (Carequality, 2025; CommonWell Health Alliance, 2025; Epic, 2025). While TEFCA is complementary to API-based exchange—providing record location and document-based retrieval at scale—it is increasingly aligned with SMART on FHIR and Bulk FHIR standards. Together, TEFCA and API-based approaches could form a dual infrastructure: TEFCA for broad, trust-based connectivity across organizations; and FHIR APIs for computable, fine-grained data access needed for clinical care, quality measurement, and AI development.
Why These FHIR Capabilities Matter for a Unified Digital and Data Architecture
Collectively, SMART on FHIR, Bulk FHIR, EHI Export, and their complementary standards constitute the minimal interoperability infrastructure required for a LHS. They reduce system-to-system variability; simplify the deployment of innovative tools; and create a stable substrate for quality measurement, value-based care, AI development and monitoring, clinical research, and patient engagement (Mandl et al., 2020; McMurry et al., 2024). Focusing on this small set of foundational capabilities—rather than expanding an ever-growing list of technical mandates—will help support data liquidity, clear investment priorities, and scalable innovation. CMS is operationalizing this alignment through its broader Health Technology Ecosystem initiative, which now incorporates the FHIR-based Beneficiary Claims Data API, Data at the Point of Care API, and the newly finalized Prior Authorization API (CMS, 2025a). The initiative also includes a National Provider Directory infrastructure and expanded consumer-access APIs so that individuals, applications, payers, and providers can securely locate provider endpoints and access health data. Together, these capabilities help form a unified digital ecosystem that enables data flow between providers, payers, and patients—laying the technical foundation for CMS’s value-based programs (CMS, 2025).
Privacy Foundations for Interoperability
As has been widely documented, the Health Insurance Portability and Accountability Act (HIPAA) is poorly aligned with modern interoperability realities (Mandl and Perakslis, 2021; McGraw and Mandl, 2021). Its de-identification provisions have become increasingly inadequate in the face of rich, multimodal data sources that make re-identification possible even when standard HIPAA safe-harbor techniques are applied (Mandl and Perakslis, 2021). Moreover, a growing proportion of health-relevant data—including data generated by consumer applications, wearables, and DTC AI tools—fall entirely outside HIPAA’s protections, leaving patients with no enforceable rights or privacy guarantees once their information moves beyond traditional covered entities (McGraw and Mandl, 2021). These gaps fundamentally limit the trustworthiness of seamless data flow and must be addressed to ensure that interoperability advances do not inadvertently increase privacy risks.
Resource Needs for Interoperability
Integrating digital health information systems into health care settings requires overcoming technical and organizational barriers such as legacy systems, limited technical expertise, and large financial investments (Boonstra et al., 2014). Effective integration demands strong stakeholder collaboration, comprehensive training, and ongoing support. Mobilizing computable biomedical knowledge involves translating clinical guidelines and evidence into machine-readable formats for integration into health IT systems, and modernizing digital health education and training is essential to prepare the health care workforce for the future (Shiffman et al., 2004; Tso et al., 2016).
However, the question of incentives underly all these challenges. Simply put, there are not sufficient incentives—financial or other—for one clinician or health system to ensure their records are interoperable so that other clinicians or health systems can access them. In fact, the inexorability of interoperability may result in patients remaining with a health care system primarily due to the difficulty of changing institutions. However, EHR uptake provides a critical lesson learned. Prior to the passage of HITECH, there was little adoption of EHRs or other digital health technologies—even despite consumer pressure for a public health record as early as the 1920s and for computerized records as early as the 1960s (Millenson, 2000; Everson et al., 2020). EHR adoption only accelerated when funding, provided by HITECH, was in place. A similar national funding push for interoperability may be necessary to see a similar shift in momentum.
Accomplishing these objectives requires a more cohesive digital and data architecture—one that enables seamless information sharing through a streamlined set of standardized functions across the entire health system, while also clearly defining the components of model systems and explicitly addressing “why” and “where” such sharing should be leveraged.
Standardized data exchange is a defining feature of an effective digital and data architecture, and the Internet offers an instructive comparison. In 1989, Tim Berners-Lee built the first website to enable document sharing among scientists (CERN, n.d.). Rather than stopping when he achieved this technical success, Berners-Lee then established the World Wide Web Consortium in 1994, standardizing concise specifications for the new system, including HTML (World Wide Web Consortium, 2025). The interoperability established by this effort and built on existing internet infrastructure rapidly sparked innovation, creating ecosystems that support commerce, search, communications, location services, and entertainment. Analogously, the widespread adoption of standardized APIs could create a similarly transformative impact on health care. Just as HTML became a foundational specification for the internet, FHIR APIs could serve as a foundational layer for health information exchange, fostering a health care ecosystem capable of continuous innovation, interoperability, and improved patient outcomes.
Digital health tools and innovations can only reach their full potential when they are built within a describable digital and data architecture. Health information that flows seamlessly between providers and patients can cultivate care that is better coordinated and uses resources more judiciously. By sharing data across settings, hospitals and primary care providers can work together to keep patients out of the hospital—for instance, through improved follow-ups after discharge to prevent readmission, facilitated by accessible digital discharge summaries and medication lists. By continuing to refine standardized approaches to accessing and sharing health data, health care is beginning to create the necessary infrastructure for a more connected, efficient, and patient-centered system. Ensuring that these advances are implemented in all care locations—from academic medical centers to community health settings—will be essential for success.
Quality Measurement and Improvement
In an ideal setting, care quality would be measured directly by health outcomes such as stroke, myocardial infarction, or major diabetes complications—data that are typically rigorously collected within clinical research environments. In routine practice, however, real-world administrative data (e.g., claims or EHRs) are commonly used as proxies to assess quality and approximate clinical outcomes. Yet these administrative measures often reflect health care system activities rather than true health outcomes, overlooking the significant influence of community factors on individual health. As data collection in an increasingly digital world expands, there are new opportunities to define more comprehensive quality metrics that capture individuals’ health throughout their daily lives.
The capacity to define, measure, and improve care quality depends fundamentally on the health data infrastructure’s strengths and limitations. A notable example of this relationship is the medication adherence quality measure, a triple-weighted metric within the Medicare Advantage Star Ratings system (Borrelli et al., 2025). Consistent medication use has been shown to improve health outcomes, decrease total cost of care, and decrease the development of medication resistance to diseases ranging from hypertension to HIV (Garcia et al., 2022). The value of improving patient medication adherence is well-established, but limitations in the ability to directly measure such adherence have incentivized low-value behaviors like using administrative data—based on paid claims for refills— as a proxy. In response to the prioritization of these measures, providers and insurers have adopted workflows promoting automatic 90-day refills, which generate continuous claims regardless of true medication use. Some have called for discontinuing this adherence measure, citing increased expenditures without clear improvements in health outcomes (DuBard et al., 2024).
Measuring quality is also particularly challenging in the US due to fragmented health data. As patients move between hospitals, nursing facilities, and primary care providers, crucial information can be lost due to data silos. This fragmentation often leads to critical missing data elements, compromising research inferences and introducing bias (Little et al., 2012). It also limits accurate measurement of clinical outcomes and fails to capture the social determinants of health. Evaluating the impact of interventions on quality thus requires improved data coordination across all care settings (Arora et al., 2022). Emerging tools, which benefit from interoperability standards and trusted data exchange, can increasingly map disparate data into secure repositories. Hosting data securely in the cloud allows powerful analytics to be run on a wide range of data sources, improving measurement of intermediate and final health outcomes. Technologies that extend measurement beyond clinical settings, such as mobile health apps that capture daily medication use, also offer promise for enhancing quality metrics compared to traditional claims-based approaches (Dayer et al., 2013).
Stronger data infrastructure will also help advance quality improvement. Although evidence-based best practices are well documented in clinical guidelines, they are rarely available as computable artifacts that can be integrated into electronic workflows. Centralized efforts in the US, such as the CMS Universal Foundation and the Healthcare Effectiveness Data and Information Set (HEDIS) Digital Quality Measure initiative are driving the development of a data infrastructure for electronic quality reporting and improvement (National Committee for Quality Assurance, n.d.). In addition to these centralized efforts, providers are also beginning to experiment with new generative AI tools to support quality improvement programs at the program level (Arora et al., 2022; Howell, 2024). Current applications are mostly task-specific, such as detecting the presence of cancer or predicting outcomes such as length of stay, readmission, mortality, or discharge diagnoses (McKinney et al., 2020). In the future, documentation processes within quality improvement workflows could also be improved to make data collection more complete and less burdensome for providers so that effort can be shifted toward analysis and driving quality improvement cycles (Howell, 2024).
Community Support of Population Health
Managing population health may now include the use of data from applications that monitor wellness, chronic disease management, or fitness (Wienert et al., 2022). As individuals may be hesitant to share such information, community advisory boards could serve as a mechanism for assisting in their collection and with analysis and implementation of improvements within that community (Guzman and Heintzman, 2024). Community-based participatory research, design thinking, and human factors research and development can encourage both incremental and evolutionary innovation and build community trust to ensure that digital health platforms truly address health disparities and access.
Accessibility
Across the landscape of digital health, accessibility is a critical yet often underappreciated component of health data infrastructure. Prior frameworks highlight that while the rapid development of cyberinfrastructure holds great promise for enhancing population and individual health, it also risks perpetuating existing inequities if not carefully managed (Shaikh et al., 2011; Badr et al., 2024). These disparities, some of which may be rooted in race, ethnicity, social class, and geography, are linked to access to communication and information resources (Badr et al., 2024).
This idea provides a foundation for understanding how digital health infrastructure, if not designed with the elimination of disparities in mind, can exacerbate those same disparities. The digital divide—the unequal distribution of broadband access and digital literacy—has been cited as a significant barrier to the appropriate use of health data to improve individual and population health outcomes, and more recent thinking includes communication technologies as important components for addressing health disparities (Benda et al., 2020; NCDIT, 2025). The Federal Communications Commission developed a comprehensive body of work on the intersection of access to broadband and health, highlighting how variation in digital access impacts health outcomes (Federal Communications Commission, 2022). The United Nations International Telecommunication Union further delineates the challenges of global digital connectivity into two primary categories: universal connectivity and meaningful connectivity (ITU, 2022). While universal connectivity refers to the basic availability of internet access, meaningful connectivity emphasizes the importance of using the internet to improve lives. This distinction underscores the need for digital health infrastructure to be both accessible and used in ways that enhance health and well-being. The Brookings Institute expands on this distinction by defining seven types of digital access necessary for bridging the digital divide: physical, financial, socio-demographic, cognitive, institutional, political, and cultural (Signe, 2023). These dimensions of access reflect the complex interplay of factors that influence whether individuals and communities can effectively engage with digital health infrastructure.
Telehealth and virtual care have become critical components of digital health infrastructure, drastically expanding remote access to essential services and health information, especially in the wake of the COVID-19 pandemic. However, the effectiveness of these technologies depends on the accessibility and interoperability of health data. To incentivize the sharing of health information by health care providers, Inland Empire Health Plan, one of the ten largest Medicaid health plans and the largest nonprofit Medicare-Medicaid plan in the United States, has taken an innovative approach: adding financial incentives for sharing health data to its quality pay-for-performance programs (Juhn and Galvez, 2022). Broadening equitable access to telehealth and virtual care will require both regulatory interventions and the development of business models that reward data sharing and collaboration among stakeholders.
There are also concrete barriers that impede the establishment of a comprehensive health data infrastructure, including outdated data strategies, regulatory barriers that cause vendor lock-in, a lack of solution architects, insufficient training, and high costs. These challenges highlight the need for a modern data strategy that prioritizes interoperability, innovation, and user-centered design. For example, patient portals that are both accessible and user-friendly can be crucial to ensure that patients can effectively engage with their health data.
Investment in more accessible and meaningful consumer- and community-centered digital health is an important step in addressing the digital divide. However, resistance to digital health tool adoption among health care providers—whether due to concerns about EHRs, workload, financial constraints, AI tools, or other issues—remains a significant obstacle (Borges do Nascimento et al., 2023). Overcoming this resistance requires a commitment to responsible use and increasing public trust, supported by policies that address information blocking, data sharing, and privacy regulations such as HIPAA. Public-private partnerships play a crucial role in this effort, and an emphasis should be placed on collective efforts in technology and training to enhance accessibility to these tools (The ACT Report, 2021). These efforts could help foster public trust, ensure compliance with data-sharing regulations, and promote responsible use in digital health infrastructure.
Direct-to-Consumer Artificial Intelligence
DTC health care models represent an emerging force within the evolving AI ecosystem, driven by large technology companies and startups that rapidly deliver AI-powered health care directly to patients (Mandl, 2025).These models bypass traditional health care organizations by leveraging extensive consumer data from social media, search behaviors, and personal devices to provide hyper-personalized and accessible health interventions. While DTC approaches offer scalability, convenience, and novel forms of patient engagement, they also pose critical challenges, including regulatory ambiguities, potential disparities in digital access, and concerns regarding data privacy and commercialization that could prioritize financial outcomes over patient welfare.
Issues for Digital and Data Architecture
As outlined above, the current lack of a comprehensive digital and data architecture results in significant barriers to delivering optimal health and health care. This gap is both costly and puzzling because in other regulated industries, digital technologies have had a more obviously and thoroughly transformative impact—raising the question of why health care has not followed the same trajectory. By improving connectivity between stakeholders and providing the means for obtaining insights from data that boost productivity and quality, these other industries have seen faster innovation over time. Crucially, the “divisions of labor” between industry, consumer groups, policymakers, and regulators get productively reformulated in ways that not only help to speed progress on technological advancements but also help align incentives across the relevant ecosystems (Lamarre et al., 2023).
What is Missing in Digital Health?
In the search for answers, there may be a strong temptation to fixate on specific technology solutions, or “technological silver bullets.” For example, one easy conclusion might be that if there was better health data interoperability technology, then the persistent siloing of health data might be relieved. But as the previous discussion on data interoperability progress reveals, new technology, on its own, will not solve health care’s problems. While there is no doubt that data interoperability—among other changes—is critically necessary to achieving a LHS, in this section the authors argue that the core issues are not merely technological, or even socio-technological, but architectural.
The concept of architecture is central to any technologically mature industry. To explain this concept, Appendix A uses analogies from three other industries—ranging from loosely- to tightly-regulated—to describe how architecture functions as a central organizing framework that encourages innovation, facilitates the alignment of incentives across the ecosystem, and accommodates continual evolution of products and services.
The Health Care Industry and its Digital and Data Architecture Immaturity
In the US, there has been a great deal of progress over the past three decades on health care digitization. At the turn of the century, some estimates state that fewer than 15 percent of patient health records were digital—and today, nearly all are (ASTP, n.d.). The instruments of health care delivery, such as medical devices, are also largely digital, including consumer-oriented devices such as smart watches and continuous glucose monitors.
Furthermore, the health technology industry and regulators have shown an increasing ability to work together. For example, over the past decade, progressive statutory pressure and federal rule-making—in parallel with the development of technology standards—have enabled patients to electronically access their health data. Over time, the data made available to patients is expected to increase and a more active data ecosystem may emerge that includes low barriers to entry, coherent addressable markets, and a well-defined foundation of government policies and open standards. Indeed, these are the beginnings of mature digital technologies and policy making capabilities, but when viewed as an industry undergoing digital transformation, the health care enterprise is far less mature than its peers in many key respects, with perhaps the most critical aspects being the following:
- Too many customized, one-off digital health infrastructure designs. While the digital infrastructure for a health care organization often has some recognizable architectural components—EHRs, admission-discharge-transfer systems, picture archiving and communications systems, and others—an industry has yet to fully form around common architecture concepts, let alone define how such “blocks” may fit together. This lack of cohesion often makes the digital infrastructure of health systems differ in crucial ways from others. Often, even neighboring departments within a single health system have incompatibly different digital infrastructures. This is not only a problem for systems management and acquisition, but also for connectivity across health care delivery, public health, and the entire health ecosystem.
- Barriers to innovation and evolution. Another consequence of the customized nature of digital health infrastructure is that the deployment of a technological innovation too often requires complex and time-consuming custom integration—which is especially challenging for new entrants into the marketplace and increases complexity, risk, and cost. Industry associations exist in several key areas of digital health, but overall, there is little momentum toward interoperability and standardization of architectural components, resulting in a lack of support that individuals receive in other, more mature industries.
- Health care leaders are unprepared for digital transformation. This discussion paper has discussed the ways in which misaligned incentives have harmed health care’s ability to harness digital health infrastructure. As an industry lacking a well-defined architecture, every stakeholder, whether large or small, is forced to “do it all themselves.” But an even more fundamental issue is that health care leaders lack an of understanding of what successful business transformation hinges on—the adoption of a digital culture throughout the organization—and the integration of digital technologies focused on meaningful customer experiences (Lamarre et al., 2023; Christou and Piller, 2024).
- Lack of aligned financial incentives. Misaligned financial incentives remain a major barrier to digital transformation in health care. Current payment models often prioritize short-term cost containment over investments in scalable, integrated digital infrastructure. This approach leads to fragmented technologies that fail to deliver meaningful interoperability or improved outcomes. Therefore, the most powerful lever for driving interoperability may be transitioning to value-based care and outcomes-based payment models. To appropriately align financial incentives, health systems should adopt value-based payment systems that reward the use of digital health tools alongside aligned public and private funding to support cohesive, long-term innovation. At the same time, as coding and reimbursement for digital health and AI tools are implemented, institutions may assume legal liability for errors or harm disclaimed by developers, further slowing uptake (Maliha et al., 2021). When provider compensation depends on coordinated care quality and patient outcomes rather than volume, the business case for seamless data exchange becomes almost self-evident. This market-driven approach could accelerate adoption more effectively than regulatory mandates alone.
The accessibility of US health data infrastructure is shaped by a complex interplay of technological, organizational, regulatory, and socio-cultural factors. Addressing these challenges requires a concerted effort to bridge the digital divide; foster meaningful connectivity; and ensure that digital health platforms are accessible, responsible, and capable of improving health outcomes for all.
The Impact of Suboptimal Digital and Data Architecture on Health and Costs
An optimized digital health infrastructure can offer transformative opportunities to address persistent challenges in the US health care system. By leveraging innovative technologies, the health care system could improve access and patient outcomes, reduce care costs, enhance patient satisfaction, and alleviate provider burnout. These advancements are vital for modernizing the system and improving America’s standing in global health affordability and outcome rankings.
To quantify the impact of improving and the digital and data health architecture, the authors considered four clinical use cases to examine how optimizing digital health data and its supporting architecture is essential to improving outcomes. These case studies are presented briefly in the sections that follow, in full in Appendix B, and summarized in Table 3, highlighting the economic challenges and opportunities for impactful reform. The authors also explore levers from the section titled “Toward a Digital-Enabled Learning Health Data Ecosystem” that apply specifically to advancing these use cases.
Use Case 1: Cardiovascular Disease (CVD)
CVD imposes substantial economic and operational burdens on health care systems. This case study describes how digital health infrastructure—such as remote patient monitoring, telehealth, and AI-driven predictive analytics—can streamline care coordination, enhance real-time patient monitoring, and improve clinical decision making. By enabling timely interventions and personalized care, these digital tools could reduce hospital re-admissions and associated health care costs and significantly improve patient outcomes.
Prioritizing the following levers could address the challenges identified in cardiovascular disease:
- Support advocacy for practical telehealth and digital infrastructure. Integrating remote patient monitoring and telehealth systems into a cohesive digital infrastructure that streamlines real-time data collection and CDS will require coordinated advocacy.
- Integrate AI-powered decision support tools. Deploying predictive analytics will assist in early detection and intervention of CVD issues, and the AI tools used in these predictions will be continuously monitored to ensure effectiveness and minimize unintended consequences.
Use Case 2: Maternal/Fetal Health and Maternal Mortality
Maternal mortality remains a critical public health concern and fragmented maternal and infant health data systems result in missed risk factors and delayed interventions. Improved digital health infrastructure—such as standardized data formats, integrated maternal-infant records, and AI-enabled predictive tools—can facilitate early detection of complications, provide better continuity of care, and improve management of pregnancy-related risks. Enhanced interoperability could reduce unnecessary medical interventions and health care costs while improving health outcomes.
Prioritizing the following levers could address the challenges identified in maternal/fetal health and maternal mortality:
- Coordinate federal regulatory standards and enforcement. Encouraging adoption of standardized data formats to streamline and coordinate maternal and infant records could help improve early detection of complications.
- Strengthen accountability mechanisms for AI development. Establishing clear AI standards and accountability for health care providers to maintain integrated maternal-fetal health records could help reduce gaps in care and missed interventions.
Use Case 3: Non-Small Cell Lung Cancer (NSCLC)
The complex nature of novel therapies for NSCLC highlights the need for precise, individualized approaches to cancer treatment. Advanced digital health infrastructure could help enable real-time monitoring of therapeutic effectiveness, streamline updates to clinical guidelines, and accelerate AI-driven multimodal predictive models. These tools could improve precision in treatment selection, safety monitoring, and patient outcomes, potentially reducing health care costs through more efficient use of resources and avoidance of ineffective treatments.
Prioritizing the following levers could address the challenges identified in NSCLC:
- Deploy strategic federal, state, and non-governmental investments. Financially incentivizing providers and health systems to implement integrated clinical pathways and advance AI-driven diagnostic tools could help ensure precision treatment selection and reduction of ineffective therapies.
- Integrate AI-powered decision support tools into clinical workflows. Gradually introducing multimodal AI predictive models into clinical workflows, supported by rigorous validation and continuous monitoring for effectiveness, accuracy, and safety could assist clinicians in choosing the best path of care for their patients.
Use Case 4: Diabetes Mellitus
Diabetes management currently involves coordinating with multiple specialists and significant patient self-management. Fragmented information systems limit effective management, leading to preventable complications and increased health care spending. Improved digital infrastructure—such as continuous glucose monitoring , telemedicine, and AI-assisted diagnostic tools—could enhance real-time monitoring, personalize patient care, and enable early identification of complications. Implementing digital health tools could help improve health outcomes, streamline care delivery, and reduce health care costs.
Prioritizing the following levers could address the challenges identified in diabetes mellitus:
- Provide comprehensive provider training programs. Expanding provider training programs in telemedicine and continuous glucose monitoring systems could help ensure that clinicians are fully equipped to integrate these digital tools into patient care plans.
- Prioritize foundational interoperability requirements. Promoting interoperable platforms that enable seamless integration of patient-generated data from continuous glucose monitors and other wearable devices directly into EHRs could help improve patient care.
Each of these use cases underscores the potential benefits of standardized, interoperable digital health infrastructures in improving clinical outcomes and reducing economic burdens across health care systems.
| Area of Waste | Source of Waste | Economic Impact | Data Sourcing Issue | Data Sourcing Solution | Digital Solution to Reduce Waste |
|---|---|---|---|---|---|
| Cardiovascular Disease | |||||
| Avoidable hospital admissions for heart failure | Lack of remote monitoring makes it more challenging to identify symptom exacerbations that lead to preventable emergency admissions | $40 billion annually in avoidable emergency room and hospital costs (Diamond and DeVore, 2022) | Heart failure metrics are inconsistently tracked across hospital records, insurance claims, and Medicare data | Distributed Data Access Platform: Links EHR data in FHIR via APIs, claims, and Medicare data sources, supporting multi-agency use and synchronization with wearable technology engaged in continuous remote monitoring | Remote Monitoring and Predictive Analytics: Uses real-time data to enable early intervention and continuous symptom tracking, reducing costly acute admissions |
| Redundant cardiovascular imaging | Routine imaging for stable patients (an average of more than one echocardiogram per year) with no change in symptoms, often performed out of caution (Van den Berg et al., 2019) | Significant imaging costs with minimal benefit | Imaging data is stored in separate systems from all other patient data, lacking integration with clinical decision records | Interoperable Imaging Repository: Provides secure access to imaging data across institutions, enabling evidence-based decision making and patient access via FHIR API | AI-Driven Imaging Decision Support: Integrates cross-agency imaging data, promoting evidence-based imaging guidelines to reduce unnecessary tests |
| Medication overuse in heart failure | On average, heart failure patients take 6.8 prescription medications and 10.1 doses a day without systematic reassessment of need and efficacy (Page et al., 2016) | Increased drug costs and potential side effects | Medication data comes from pharmacy records, EHRs, and payer data, but lacks synchronization | Integrated Access to Pharmacy and Claims Data: Connects pharmacy, payer, and EHR data to enable real-time monitoring and patient access via approved applications | Digital Medication Management: AI-driven assessment tool continuously evaluates prescriptions across sources, optimizing efficacy and safety |
| Maternal/Fetal Health and Maternal Mortality | |||||
| Missed prenatal screenings | High-risk pregnancies lack comprehensive screening, with 2.6 greater odds of missed screening for racial/ethnic minorities, leading to undetected conditions and preventable complications (Stegman et al., 2023) | Missed prenatal screening leads to 13 percent greater rates of congenital heart disease detection (Pinto et al., 2014) | Screening data for maternal health may differ between administrative reports, public health registries, and hospital EHRs, creating redundancy and gaps | Commonly Sourced Delivery System Data for Administrative Reports, Registry and EHR Notification System: Connects public health registries, hospital EHRs, and patient applications to provide seamless screening alerts for providers and patients | EHR-Based Screening Protocols with Population Health Alerts: Automated reminders and alerts for high-risk pregnancies aligned with federal standards to ensure timely screenings, interventions, and risk-appropriate care |
| Fragmented maternal and infant care | Disconnected records lead to redundant tests, undiagnosed conditions, delays in care, and inadequate follow-up | Increased health care costs and risk of adverse events like maternal, fetal, or infant morbidity and mortality | Maternal, fetal, and infant records are often stored as separate EHRs despite a linked birth hospitalization event, which may result in loss of important information for the dyad | Longitudinally Linked Maternal-Infant Record System: Virtually links maternal and infant records across institutions and over time, supporting access via patient portals | Continuity of Care System: Enables sharing of holistic health history for timely care and reduced redundancy, allowing families secure access to records and helping clinicians provide more consistent quality care |
| Preventable emergency deliveries | Absence of early risk monitoring may result in emergency interventions for preventable high-risk pregnancy issues | Emergency deliveries often result in NICU admissions or C-sections, which may cost 50 to 100 percent more than vaginal delivery (Valencia et al., 2022) | Emergency room, urgent care, and postpartum readmission data are collected differently across Medicaid, hospital quality measures, and state health registries | Postpartum Monitoring Access: Integrates data across Medicaid, state, clinic, hospital, and home to allow for comprehensive patient monitoring | Remote Monitoring and Telehealth for High-Risk Pregnancies: Patient-centric monitoring (e.g. home blood pressure monitoring) detects risks early, helping to reduce emergency costs |
| Non-Small Cell Lung Cancer | |||||
| Delayed cancer diagnosis | More than 90 percent of patients experience a 5 to 6 month delay in diagnosis and only 4.5 percent of eligible individuals were screened in 2022 (Gildea et al., 2017; Sonawane et al., 2024) | Treating advanced stage cancer costs more than earlier stage cancer. Also, approximately $39 billion in productivity has been lost due to lung cancer (Bradley et al., 2008). | Cancer diagnosis data is split across the Surveillance, Epidemiology, and End Results Program (SEER), EHRs, and state registries, with inconsistent timing and methods (NIH, 2025) | Cross-Registry Cancer Data Access: Connects SEER, state registries, and EHRs, supporting multi-agency data use and patient retrieval of screening history | Population Health Screening Programs with EHR Integration: Prompts regular screenings and follows up for high-risk populations through interoperable platforms |
| Insufficient early cancer diagnosis | Only 27 percent of lung cancer patients are diagnosed at early stages, when prognosis is better. Also, there is inadequate genetic testing to identify driver mutations (American Lung Association, 2024). | High cost and 85 percent lower 5-year survival rate with late-stage treatment compared to earlier stage treatment (Cancer Research UK, 2022) | Screening rates are inconsistently captured in SEER versus Medicare and other health records | Interagency Screening Alerts: Connects SEER and Medicare data to enable timely, consistent alerts that are accessible to providers and patients | AI-Enhanced Diagnostic Alerts: Uses AI-powered alerts through EHRs to prompt screenings and follow-ups, improving early diagnosis rates |
| Lack of access to high-quality cancer care | 21 percent of lung cancer patients receive no treatment, and socioeconomic and access barriers contribute to this gap (David et al., 2017) | Increased mortality and health care disparities | Access data is variably tracked by EHRs, claims data, and community health reports | Access Coordination Hub: Links EHR, claims, and community health data for coordinated outreach via integrated applications | Community-Based Digital Outreach Programs: Leveraging integrated digital records, this program would support outreach to high-risk patients and increase care access |
| Overuse of aggressive cancer therapies near the end of life | High rate of medication without timely reassessment, leading to unnecessary palliative expenses | 2.5 times greater costs for patients using chemotherapy near the end of life (Garrido et al., 2016) | Palliative care metrics are collected differently between hospital records, palliative care centers, and insurance claims | Shared Palliative Care Database: Links data across hospitals, palliative centers, and insurers, and patients can access their care plans through health applications | AI-Driven Palliative Care Assessment: AI-based reviews informed by distributed palliative care data ensure treatment aligns with current needs |
| Diabetes Mellitus | |||||
| Poor glucose control among diabetics | Infrequent checks and suboptimal adherence to finger stick and hemoglobin HA1c monitoring can lead to insufficient routine glucose level monitoring | Increased rate of diabetes-related complications and increased expenses for glucose monitoring supplies | Lack of real-time glucose monitoring and of integration of point of care or home-based glucose readings with the EHR | Home Glucose Data in Health Records: Linked home-based glucose monitoring data with the EHR | Tracking Glucose in Real Time: Continuous glucose monitoring allows real-time tracking of glucose to avoid need for finger sticks and facilitate personalized dietary recommendations |
| Excessive end-organ complications from diabetes | Inaccurate and delayed ophthalmology screening leads to preventable progression and detection of diabetic retinopathy | Preventable blindness and related expenses | Screening rates and accuracy are variable between primary care providers | Automated Referrals to Eye Specialists: Linking algorithm output with EHRs to facilitate automated referral to retinopathy specialists | AI-Based Eye Screening: Deep learning-based diabetic retinopathy screening can improve accuracy and automation of retinopathy screening |
| Underutilization of diabetes self-management programs | Poor self-management of diabetes medications and lifestyle changes lead to poor glucose control | Increased rate of diabetes-related complications | Lack of wireless and broadband capabilities, particularly in rural areas, makes telemedicine-based solutions challenging | Telemedicine Tools for Diabetes Self-Management: Facilitating access to high-quality telemedicine platforms to reinforce diabetes self-management strategies | Telemedicine-Based Diabetes Prevention Programs: Telemedicine platforms facilitate diabetes self-management, providing comprehensive, patient-centered care that extends beyond traditional clinical settings |
Table 3 | Digital Solutions to Reduce Waste in Health and Health Care: Four Primary Use Cases in Cardiovascular Disease, Maternal/Fetal Health and Maternal Mortality, Non-Small Cell Lung Cancer, and Diabetes Mellitus
SOURCE: Developed by authors.
The financial and care-related outcomes of the US health care system are increasingly dependent on the widespread adoption of digital health technologies. As demonstrated in use cases like those described above and in Appendix B, the failure to integrate and scale these capabilities will likely not only lead to poorer health outcomes but also escalate costs. By embracing solutions like a robust digital and data architecture, health care can shift from a reactive, costly system to a more proactive, efficient one that improves patient outcomes while reducing long-term health care expenditures. Addressing these issues will require the strategic utilization of financial, legal, regulatory, and cultural levers to deliver long-term value while improving health outcomes for all.
Toward a Digital-Enabled Learning Health Data Ecosystem
Digital infrastructure for health has advanced tremendously over the past three decades. And yet, as this discussion paper has highlighted, the lack of connectivity and alignment across the health ecosystem is a key factor in health care’s failure to reduce needless human and financial costs. At a high level, the benefits of digital technologies are clear. There are valuable insights and critical efficiencies to be extracted from digital data, all while improving access to care, reducing waste, supporting the health care workforce, and achieving a system that continuously learns from individual-level and population-scale experiences. Unfortunately, in practice, the health care system shows a stubborn resistance to such effective transformations—even if the underlying industry of health technology infrastructure was mature enough to support them, which it is not. In fact, the pace of innovation and technological evolution—as well as economic, legal, policy, cultural, and regulatory levers—are both slower and less effective than peer industries (Abernethy et al., 2022).
Forming, reforming, and maturing an industry is a daunting prospect. In the home construction, global telecommunications, and global financial industries—discussed in Appendix A—the maturation process was far from orderly and linear. However, transformation of the digital health industry, in one form or another, is more or less inevitable. Human endeavors are becoming increasingly technological, and there is no likely future in which digital health escapes this trend. Thus, the central question is this: “How must we intervene, in this moment, to guide the maturation of the digital health technology industry in the most beneficial way?”
A Phased Approach
While there are many factors at play, including several distinct challenges that will need to be addressed, this discussion paper specifically focuses on the need, as a first step, to have a broad and common understanding of the importance of digital and data architecture. To achieve this understanding, convening stakeholders—including health care systems and their leaders, public health professionals, front-line health workers, policymakers, patient advocacy groups, and technology companies—will need to instigate workstreams and develop processes that bring the entire industry together to find common understanding and agree on goals.
A phased approach can allow for steady progress towards the ultimate goal. This section presents a proposed phasing plan that can serve as a starting point for discussion, noting that the items outlined below do not need to be performed in a linear fashion:
- Stakeholder convening. Gather key players from health care, patient and community advocates, technology, public health, and policy to advance the understanding of digital and data architecture; learn from other industries; build on federal, state, and local initiatives; and establish a common vision and goals.
- Framework development. Create collaborations to draft initial architectural blocks, defining their functional roles in enabling a LHS and high-level guidelines for interoperability, privacy, and security.
- Pilot programs. Support the launch of small-scale projects to test and refine key elements of the framework in real world settings.
- Feedback and iteration. Collect data and insights from the pilot programs to improve standards, develop metrics for outcomes and costs, and identify key challenges.
- Scaling and integration. Create and encourage incentives to expand successful pilots and integrate them into health care organizations.
- Policy advocacy. Convene policymakers, regulators, health care and public health leaders, and framework developers to identify ways to embed the components of the framework into regulations and incentivize broader adoption.
- Infrastructure investment. Secure investment, potentially through government channels, to fill market gaps and incentivize necessary digital infrastructure.
- Education and training. Develop programs to train health care professionals on the use of new digital technologies.
- Public engagement. Inform and involve patients, community leaders, and the public to build trust and encourage adoption.
- Ongoing innovation and adoption. Regularly update the framework to accommodate new insights and technologies, ensuring that it remains flexible. Furthermore, develop and regularly update a roadmap outlining future digital infrastructure goals so that all stakeholders have insight into how the digital infrastructure will evolve.
- Evaluation and monitoring. Continuously measure the impact of digital tools and the larger digital and data architecture on patient outcomes, cost reduction, access, and overall health quality to assess how well the support for a LHS is being delivered.
- Global alignment. Collaborate with international organizations to align standards globally, driving innovation and enabling cross-border health care and public health interoperability.
By setting realistic and achievable milestones within each phase and prioritizing coordination and collaboration between technology, health care, public health, patient, and policy stakeholders, expanding an understanding of digital and data architecture can be approached in more manageable chunks.
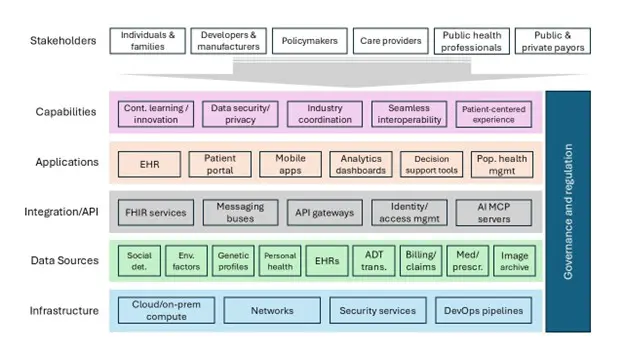
Figure 2 | Health Digital and Data Architecture Conceptual Framework
SOURCE: Created by authors.
NOTE: ADT = admission, discharge, and transfer; Cloud/on-prem compute = Cloud and on-premises computing; MCP = model context protocol; Med/prescr. = medication and prescription; Pop health mgmt. = population health management.
Defining a health digital and data architecture and then organizing an industry around it will be a major long-term effort. As a starting point for discussion, Figure 2 presents a proposed health digital and data architecture. Structurally, this proposed digital and data architecture takes the form of a “technology stack”. If using home construction as an analogy:
- Capabilities = Choices regarding comfort, aesthetics, etc.
- Applications = Building functions (heat retention, physical safety, etc.)
- Integration/API = Building components (framing, roofing, plumbing, insulation, etc.)
- Data sources = Building materials (lumber, nails, pipes, tiles, etc.)
- Infrastructure = Raw materials (wood, steel, concrete, stone, etc.)
A digital health infrastructure industry could also be organized along these five layers, and governance—like technology evolution and interoperability—could be focused on specific areas of relevance, like happens in other regulated industries.
Dealing with Inertia Through Bold Action
It is important to identify the organizations best positioned to lead and coordinate the activities needed to advance this digital and data architecture. However, sustaining meaningful progress may necessitate creating specialized entities or adapting and expanding the mandates of existing organizations to guide this effort in the long-term. Government engagement will also be pivotal—ideally in an observer and facilitator role—to assist in tracking progress, identifying critical gaps, and strategically applying policy interventions without directly driving implementation. Ultimately, achieving substantial progress in digital health depends on robust coordination across the entire health and health care ecosystem.
Health industry incumbents already have a stake in the current system and may resist changes that could disrupt their market positions. This situation is not entirely unreasonable—much like a subcontractor on a home building project who, without a clear architectural plan, cannot easily coordinate with other subcontractors, today’s health technology vendors often manage highly customized end-to-end deployments with little integration with their peers. This dynamic frequently leads to the creation of “walled gardens,” which not only safeguard the vendors’ market position but also discourage new entrants and hinder interoperability. Thus, it will be critical to find ways to overcome this kind of resistance while still encouraging needed investment. Having said that, incumbents typically stand to benefit the most from a more standardized and modular system. For example, in the banking industry, there was resistance to digital transformation due to fears that it would reduce foot traffic in physical banks. However, institutions that embraced online banking and mobile applications saw immense benefits through increased customer engagement, lower operating costs, and improved services. A similar dynamic is playing out today in the realm of digital currencies (Kumar et al., 2024).
In digital health, embracing technological advancement can help incumbents streamline operations, enhance patient satisfaction, and remain at the forefront of health care innovation. Cutting across these arguments are overarching drivers that have caused misalignment within the health system, including systemic barriers, mistrust, uncoordinated financial incentives, and a lack of data infrastructure. Overcoming incumbent inertia in a health technology industry with such indirect consumer pressure may require the use of financial, regulatory, legal, and cultural interventions to gain the leverage needed for greater system alignment.
In this section, the authors examine the drivers that have caused health system misalignment and provide a menu of possibilities that highlight potential legal, regulatory, industry, and other levers that can mobilize collective efforts and define long-term strategies to realize the envisioned digital and data architecture described in this discussion paper.
Misalignment Driver: Regulatory Complexity
The lack of consistent and streamlined policies and regulatory frameworks in digital health—particularly around EHRs—create significant barriers to aligning financial incentives, data standards, and compliance measures. Overly complex and fragmented regulations prevent prioritization of core interoperability components, undermining progress toward a cohesive digital infrastructure. Without a clear and sustained commitment to simplification and alignment with the protocols outlined in the 21st Century Cures Act, the development of a LHS will likely remain hindered by regulatory inefficiency.
Levers That Can Be Used to Address Regulatory Complexity
- Prioritize a small set of foundational interoperability requirements.
- Regulation could focus on a concise set of high-value, standards-based capabilities—SMART on FHIR for individual access, Bulk FHIR for population-level exchange, and EHI Export for full-record portability—while beginning to sunset certification requirements unrelated to interoperability. Concentrating oversight on those core functions and allowing most other EHR functionality to evolve through market competition and innovation could reduce regulatory burden and direct investment toward infrastructure that enables computable data exchange across settings.
- Align federal agencies to create a coherent regulatory environment.
- Coordinated action across CMS, ASTP/ONC, OCR, FTC, and FDA could harmonize privacy protections, patient access rights, market fairness, and technical interoperability expectations. A unified federal posture could reduce duplicative or conflicting requirements, create predictable regulatory signals, and give providers and developers confidence to modernize their systems around a shared, scalable, and interoperable digital and data architecture.
- Empower patients to acquire, use, and share their digital data.
- Interoperability initiatives can improve patients’ ability to obtain complete, computable copies of their information—including multimodal data like clinical notes, images, device-generated measures, and -omics data—and share that information seamlessly with providers, researchers, public health agencies, and DTC applications. Enabling patient-directed data exchange could strengthen self-management, care coordination, and discovery; improve population health; and support a digital ecosystem in which individuals can choose the tools and services that best promote their well-being.
- Promote complementary interoperability standards.
- To maximize the utility of core APIs, policymakers could encourage the adoption of complementary event-based standards like FHIR CDS Hooks and FHIR Subscriptions, which support workflow integration, real-time clinical notifications, and decision support triggers. Cultivating these standards could strengthen the broader interoperability ecosystem without expanding prescriptive functional mandates on EHR products.
- Incentivize and measure interoperability performance.
- Linking reimbursement, reporting, and administrative simplification to the effective use of standardized APIs reinforces high-performing exchange rather than checklist compliance. Emphasizing outcomes like timeliness, completeness, and usability could help promote market-driven improvements in user experience—like eliminating redundant manual data entry—and could support quality measurement, value-based care, AI development, and public health applications.
Why it Advances the Digital and Data Architecture
By strategically focusing regulation on a limited set of proven interoperability requirements, policymakers could reduce regulatory complexity and provide clear investment signals for health care organizations. This targeted approach would channel resources toward high impact, widely adopted capabilities that support a coherent and scalable interoperability digital and data architecture. As these foundational APIs continue to mature, they can also enable progress across multiple priorities, including quality improvement, value-based care measurement, AI development and monitoring, regulatory oversight, and large-scale clinical research. Progress in these priorities could subsequently strengthen the digital infrastructure that underpins a LHS and supports sustained innovation.
Misalignment Driver: Industry Fragmentation
The health care technology industry’s disorganization makes it nearly impossible for health care providers and payers to adopt interoperable systems despite growing patient demand. This dysfunction prevents meaningful progress; limits patient access to their own data; and undermines efforts to deliver coordinated, high-quality care, perpetuating inefficiencies and inequities.
Levers That Can Be Used to Address Industry Fragmentation
- Coordinate public awareness and messaging frameworks.
- National campaigns led by public health organizations, patient advocacy groups, and professional societies could highlight the critical role of digital health in improving care quality, transparency, and access. These campaigns could emphasize how a digital and data architecture benefits patients, providers, and the broader health care ecosystem. Messaging could include real-world examples—such as how interoperability can help reduce redundant testing or how AI-powered tools can improve early detection of diseases—to make the value tangible to patients and other stakeholders.
- Leverage consumer demand for data access.
- Building on public awareness campaigns that highlight the benefits of digital health, empowering consumers and patients to demand seamless and direct access to their own health data could significantly drive change. Consumer-facing initiatives led by public health organizations, patient advocacy groups, professional societies, or others could actively educate and promote the demand for personal health data access. This education could create crucial market pressure on vendors, developers, and providers to adopt truly interoperable systems that enable individuals to own and control their own data.
- Strengthen accountability mechanisms.
- HHS could establish and enforce robust accountability mechanisms to safeguard data privacy, ensure quality, and promote patient safety in health care delivery. By collaborating with compliance organizations and quality assurance networks, HHS could maintain high clinical standards while consumer protection advocates push for stricter measures to protect patient interests, particularly in high-risk areas. Adopting and enforcing stringent security protocols and data permissioning frameworks could help ensure that sensitive health information is protected while allowing appropriate access for care delivery and innovation.
- When the financial sector introduced interoperability mandates through International Organization for Standardization (ISO) standards, it directly accelerated global adoption of secure digital transactions. Similarly, enforcing interoperability standards in digital health through regulatory penalties could catalyze rapid industry adoption. Health information technology vendors whose products fail to meet interoperability requirements—like compliance with FHIR standards—could be subject to penalties, including fines and revocation of certifications mandated by ASTP/ONC. These certifications are essential for vendors seeking to sell EHR systems to health care providers participating in federal health care programs including Medicare and Medicaid and therefore would be persuasive penalties.
- Develop a flexible regulatory framework for AI systems.
- Just as global telecommunications standards like 4G and 5G enable seamless international connectivity, clear enforcement mechanisms on interoperability standards could help ensure a similarly cohesive infrastructure across health care. A regulatory framework could require routine audits of AI diagnostic systems to ensure adherence to safety, quality, and interoperability standards. These audits could provide transparency and foster trust in AI-driven health technologies. For example, certification programs could evaluate and label AI systems for safety, fairness, and interoperability, providing stakeholders with clear guidance on reliable tools while ensuring alignment with existing infrastructure.
Why it Advances the Digital and Data Architecture
Such enforcement mechanisms could require health technology vendors to go beyond simple data sharing and interoperability mandates and make progress towards adopting a parsimonious set of carefully chosen digital and data architecture and interoperability standards. These enforcement mechanisms would encourage, for instance, EHR vendors to participate in industry-wide efforts to define digital and data architecture standards and penalize those who refuse or fail to share patient data appropriately. Regulatory penalties for non-compliance would directly target vendor lock-in and data fragmentation by ensuring that vendors adhere to interoperability digital and data architecture standards, much like building inspectors ensure compliance with structural codes. If coupled with reimbursement incentives for health care providers to procure new technology, these enforcement mechanisms could help align incentives for vendors to prioritize interoperability, transparency, and compliant data exchange.
Misalignment Driver: Misaligned Financial Incentives
Insufficient investment from government agencies, non-governmental organizations, and public-private partnerships hampers the development of critical infrastructure, digital innovation, and research. This lack of resources limits the ability to build scalable systems and improve care outcomes, particularly for underserved populations.
Levers That Can Be Used to Address Misaligned Financial Incentives
- Encourage strategic federal, state, and non-governmental investments.
- Federal agencies, state agencies and non-governmental organizations like philanthropies can play complementary and collaborative roles in strengthening health care infrastructure. Federal and state funding could prioritize the development of interoperable data sharing frameworks, research programs, and systemic quality improvements, while philanthropy could focus on preventive health initiatives, patient education, and community resource enhancement.
- Build national infrastructure for a robust digital and data architecture.
- ASTP has made important progress in advancing standards and governance frameworks to support a national digital and data architecture. However, a dedicated authority, additional resources, and cross-agency coordination is likely needed to harmonize investments and sustain capacity to evolve the digital and data architecture alongside emerging data standards and use cases.
- Empower providers and patients through digital literacy education.
- Universities, academic medical centers, and professional societies could develop and implement training programs focused on digital health literacy, ethical data management, and patient engagement. These initiatives could help prepare health care professionals to effectively adapt to using digital tools, fostering a workforce equipped to seamlessly integrate technology into patient care. For example, a health system could collaborate with an academic institution to develop an interdisciplinary curriculum for clinicians, IT staff, and administrative leaders. The program could emphasize data ethics, interoperability standards, and practical applications of digital tools in care delivery.
- Public health organizations and patient advocacy groups could also spearhead educational campaigns to improve digital health literacy among patients and families. By helping individuals understand and use their health data, these efforts could help ensure that both patients and providers actively engage with digital health tools, enhancing the overall ecosystem.
- Establish federal workforce development programs.
- Federally-funded mechanisms—examples including National Institutes of Health (NIH) F-series fellowships, T-series institutional training grants, and mentored K-series awards (like K12 and KL2) that cover both individual investigators and institutional programs—offer protected time, mentorship, and research support for PhD trainees, postdoctoral fellows, and early-career faculty. Aligning these programs with modern digital health competencies could help cultivate a workforce capable of translating advanced analytics into clinical practice, accelerating innovation and spreading best practices throughout health systems.
Why it Advances the Digital and Data Architecture
Government and philanthropic partnerships could drive the creation of interoperable, open-source platforms adaptable across the full range of care settings. These efforts could help reduce health disparities, promote innovation, and contribute to a unified and patient-centered digital ecosystem. Strengthening workforce capacity through targeted education could help ensure that health care professionals and patients are not just passive users but active participants in the digital health ecosystem. This grassroots engagement could help drive adoption, innovation, and integration of digital solutions into everyday care.
Misalignment Driver: Resistance to Innovation
Resistance to adopting new standards and AI systems often stems from the perception that such changes will be enormously disruptive. Phased approaches, such as pilot programs and gradual integration, coupled with effective monitoring, could help individuals and organizations see the value and feasibility of these innovations, speeding adoption and progress in improving systems and outcomes.
Levers That Can Be Used to Address Resistance to Innovation
- Promote integration of AI-powered decision support tools.
- Health systems can prioritize the deployment of AI tools for CDS, including early detection algorithms and risk stratification models. These tools should, when deployed, be integrated into existing workflows, ensuring they augment rather than burden clinical decision making.
- Encourage the adoption of federated systems for collaborative AI development.
- Encouraging the adoption of federated approaches would allow multiple organizations to train AI models collaboratively without sharing raw data, accelerating innovation while maintaining strict data privacy (McMurry et al., 2024).
- Establish AI validation and monitoring protocols.
- Establishing algorithmovigilance programs for AI tools could assist patients and providers in understanding how algorithms meet safety, fairness, and performance standards before and after deployment. Criteria for these programs could include effectiveness thresholds, bias mitigation, explainability, standards-based monitoring, and explainability, fostering stakeholder trust in emerging technologies. Certification paradigms could be considered as well.
Why it Advances the Digital and Data Architecture
Emerging technologies and AI could help drive efficiency, accuracy, and scalability in health care delivery. These levers could help ensure that innovation aligns with security, privacy, safety, and interoperability goals, enhancing the overall digital and data architecture while addressing the industry’s most pressing challenges.
When contemplating investment priorities for a healthy nation, acknowledging potential resistance and showing how a phased, inclusive approach can benefit all stakeholders could help ease concerns. Utilizing approaches that fill gaps, encourage investment and risk-taking, and motivate willingness to change are also critically important. All those involved in advancing health and health care should be prepared to learn from what has worked before, while ensuring the boldness and creativity necessary to envision and try new approaches.
Marshaling the Will
Technology alone will not fix US health care. Instead, technology should serve as an enabler of broader systemic improvements. The conversation must shift toward leveraging technology to advance clearly defined objectives such as reducing administrative burdens, improving equitable access, facilitating better patient outcomes, and enabling continuous learning and improvement. Successful examples from other industries show the need for a foundation of strong policy frameworks, universal care access, and strategic investments in primary care and preventive measures—placing technology in a supportive rather than central role.
Improving US health care will require integrating technology into a coherent, well-designed health system where policy, culture, and infrastructure are aligned. A parsimonious approach to interoperability, centered on a common digital and data architecture and supported by aligned financial incentives, is critical for ensuring this alignment. Such an approach ensures a focused investment on infrastructure while enabling innovation. A robust digital and data architecture plays a fundamental role in moving toward this functional system by establishing a robust yet efficient foundation that aligns technology solutions, policy directives, and cultural practices into a cohesive, effective whole.
Transforming health care’s digital and data architecture requires the coordinated application of multiple policy and market levers, each addressing different barriers to progress. Incentives should be realigned to reward interoperability rather than information blocking and system lock-in. Better governance and clearer authority should accelerate standards development and implementation. Workforce capacity should expand through targeted educational investments and clearer career pathways for health informatics professionals. Enhanced transparency and practical control mechanisms for personal health information should increase patient empowerment and involvement. Finally, these levers should be applied with sensitivity to the varied starting points of different health care organizations, creating pathways for progress regardless of an organization’s size, resources, or technical sophistication.
The time is now. Technology is advancing. The question is not whether digital transformation will occur—it is essentially inevitable. Left on its own, this transformation is likely to lead to a digital and data infrastructure that is even more wildly complex, highly fragmented, and siloed than it is today. Instead, we can intervene now and organize the health care industry and regulatory levers around a digital and data architecture to create a better health future for all.
The data, technology, and expertise to transform care exists, and yet, fragmented systems, misaligned incentives, and outdated policies stand in the way of realizing digital health’s full potential. The path forward is clear: establish a trusted and robust digital and data architecture that protects patients, ensures equity, and accelerates innovation. No single entity can achieve this alone. Collaborative leadership across sectors is necessary to build the infrastructure, standards, and social license that will enable seamless and secure digital benefits that can turn information into insights and insights into healthier lives for all. The moment for coordinated national action is here to catalyze the progress individuals and communities deserve.
Appendix A
Comparison to Other Regulated Industries
Improved coordination, regulation, innovation, and incentives alignment are characteristics of a mature digitally transformed industry—one that is organized around well-accepted sets of architectural concepts. Architecture gives meaning to technology. When considering the technology of modern data standards for health data interoperability, standardized data elements are akin to standardized building materials in the home construction industry. But standardized data elements or building materials, without an overarching concept of architecture, do not make for a mature industry. In essence, architecture explains why and how interoperable lumber, pipes, and wires are needed. The authors of this discussion paper believe that the same is true with respect to data standards in health care.
To illustrate this, the authors highlight the similarities and differences between the current state of the health care industry and three mature industries that operate within robust architectural frameworks: home construction, global telecommunications, and global investment banking.
The Home Construction Industry
Home design is organized around the concept of architecture—and so is the industry of home construction. The home construction industry is composed of orderly concepts such as foundations; framing; plumbing; heating, ventilation, and air conditioning (HVAC); electrical systems; roofing; doors and windows; and so on. Each of these components defines a sub-industry “block” with its own standards and practices around materials, labor, technology, and markets.
Architecture provides an organizing framework that reduces coordination complexity and encourages the development of useful interoperability standards. To understand this, consider that when building a house, a general contractor will issue subcontracts to a roofer, plumber, carpenter, electrician, and so on. Critically, each subcontractor can then proceed with drastically reduced coordination. Without the architectural organization, unwieldy coordination amongst and between each subcontractor would be required. Furthermore, these architectural concepts enable the industry to focus interoperability requirements more productively. The physical requirements for how the pieces of a house should fit together become simplified, and the associated sub-industries have clarity on how to organize and work together towards productive standards.
Home building industry associations often collaborate with regulators to define relevant building codes, again organized around architectural components (Potter, 2022). This leads to productive regulatory interfaces between industry and government. Roughly speaking, it puts the onus on the industry to help define requirements, including evolving them as new technologies become available, and then the role of regulators is to assess whether these requirements ensure safety and are faithfully adhered to.
Architecture also helps to encourage technological innovation. Imagine that an inventor developed a new type of HVAC technology. Since widely used architectural standards exist, the innovation can be brought to the home building market in a standardized way. Without architectural standards, a new technology to keep a home’s inhabitants warm would likely involve customized design and deep system integration into each and every home building project—a prospect that is sadly common today when integrating new technology into disparate health care systems, and which in practice is inefficient for innovators trying to build successful businesses.
Finally, architecture facilitates the alignment of economic and other incentives. In effect, the conceptual “blocks” help the industry to be better organized and reward innovation, specialization, coordination, and regulation. Indeed, it is not just the houses that are organized around architecture—so are home building industry associations.
The Global Telecommunications Industry
Today, a person can travel to nearly any well-inhabited place in the world and use a mobile digital device to connect to nearly anyone, anywhere, at any time. The architecture of the global telecommunications system that makes this possible is based on architectural concepts such as transmission media (fiber optics, satellites, microwave links); network infrastructure (cell towers, internet exchange points); protocols (transmission control protocol/internet protocol, 4G/5G); service providers (internet service providers, telecom providers); end-point devices (smartphones); and so on. Besides global connectivity, these architectural elements support the system’s evolution and facilitate the adoption of new technologies, including interaction with regulatory authorities in each part of the world.
When new technologies emerge, industry associations such as the Institute of Electrical and Electronics Engineers, the International Telecommunications Union, and the 3rd Generation Partnership Project work to ensure compatibility with global infrastructure (3GPP, n.d.; IEEE, n.d.; ITU, n.d.). But the focus on ensuring a robust and interoperable architecture by these industry associations achieves more than seamless interconnectivity—it also encourages and supports component-level and global innovation and evolution.
Cisco Systems, for example, started as a small innovator developing new routing technologies like the Enhanced Interior Gateway Routing Protocol that could handle multiple network protocols (Cisco Systems, 2023). By marketing their innovation as a superior replacement for existing “blocks” in the global telecommunications architecture, Cisco addressed the entire telecommunications market and avoided costly one-network-at-a-time integration. Through strategic partnerships, acquisitions, and continuous innovation, they became a market leader (Statista, 2024). More recently, innovations such as low-earth orbit satellite communications networks like Starlink create new opportunities while integrating into existing global markets (Starlink, n.d.).
Industry-wide, global evolution is also organized around architecture. The transition from 4G to 5G, for example, focused on transmission media such as New Radio, network infrastructure that supports network slicing for applications such as massive machine-type communication, advanced protocols for improved security, and endpoint evolution—making it possible to coordinate such fundamental technology evolutions in an organized manner (Dutkiewicz et al., 2017; Arfaoui et al., 2018; Vook et al., 2018).
The Global Financial Industry
Today, a person can walk into a store in most parts of the world and use a credit card to make a purchase. The global financial architecture, composed of card networks, banks and acquirers, merchants, payment gateways, point-of-sale systems, and clearing and settlement systems make this possible. These components work together to create a robust and efficient system that allows for the global use of credit cards by consumers, guaranteed receipt of payment by merchants, and profitable transaction processing fees for banks. These components also ensure that transactions are processed quickly, securely, and privately, while still allowing for regulatory oversight and law enforcement.
Industry associations such as ISO and the Payment Card Industry Security Standards Council bring the industry together to define interoperability and security standards organized around the architecture of global financial services (ISO, n.d.; PCI Security Standards Council, n.d.). The Financial Industry Regulatory Authority, in coordination with counterparts in other countries, began as a conference of financial organizations that emerged from the economic calamity of the Great Depression to work together on ensuring market integrity and investor protection (FINRA, n.d.). Formed as an industry association, it eventually became a productive interface between industry and government regulatory organizations like the Securities and Exchange Commission (SEC, n.d.).
When technological innovations such as digital currencies emerge and questions about the possibility of central bank digital currencies are raised, the technological evolution, policy recommendations, and regulatory and political implications are studied in terms of the impact on the global financial system’s architecture. New concepts—like the XC platform—can be defined as multilayer architectures for financial settlements that make use of new technology concepts like digital certificates of escrow to accommodate digital currencies in a way that harmonizes with the rest of the architecture, flexibly adapting to new innovations (Kumar et al., 2024).
A major function of this architecture, besides providing seamless global interoperability, is the prevention of financial fraud in a way that balances privacy with the need for market security and stability. The global financial system’s architecture enables the use of technologies like machine learning (ML) in targeted ways to detect fraudulent activity and while remaining fully isolated in payment processing or clearinghouse systems to aid in managing consumer privacy. The interoperability between banks and card networks provides an alignment of incentives as they both realize significant financial rewards through any improvements in privacy preserving detection algorithms since merchants are willing to participate in exchange for assured payments.
Appendix B
Case Studies to Illustrate Costs and Opportunities in Digital Health Data and Infrastructure
Use Case 1: Cardiovascular Disease
CVD is the leading cause of mortality worldwide, accounting for nearly one-third of all deaths (Martin et al., 2024). The burden of CVD—including heart attacks, strokes, heart failure, and amputations—poses significant challenges to health systems due to its high prevalence and associated costs. The integration of digital technologies into established CVD care, such as AI-driven predictive analytics, telehealth, and remote patient monitoring, offers transformative potential to address these challenges (Cleland et al., 2005; Chow et al., 2015; Santo and Redfern, 2020). By enabling better care coordination, proactive care, cost efficiencies, and improved discovery of disease phenotypes and treatments, digitally-informed care for CVD could improve patient outcomes, reduce hospital readmissions, and lower overall health care costs.
CVD and the Need for a Digital Health Infrastructure
Heart failure, or cardiomyopathy, is a cardiac condition that impairs the heart’s ability to contract and relax, resulting in circulatory insufficiency and resultant fatigue, fluid retention, and higher risks of cardiac arrhythmias (American Heart Association, 2024). It is one of the most prevalent cardiac conditions in the US, affecting 6.7 million adults (Martin et al., 2024). It is typically a chronic condition, and its management is complex for both clinicians and patients, involving a variety of lifestyle, pharmacologic, and device-based interventions (Maddox et al., 2024). Specifically, patients are often required to adhere to a somewhat restrictive diet; carefully manage their hydration; and engage in healthy patterns of exercise, sleep, and activity. Depending on the degree of cardiac impairment, patients may need to take 10 to 15 medications a day and use cardiac devices to monitor and manage abnormal heart rhythms. Their care team typically consists of several physicians—primary care, general cardiologist, heart failure specialist—and a variety of other care team members, including nurses, physical therapists, dieticians, social workers, and pharmacists. Patients often need to juggle many tasks simultaneously to optimally manage their condition, including monitoring their heart status with frequent vital sign and weight checks, coordinating multiple prescriptions, communicating with several care team members, and attending a variety of ambulatory care visits. Care teams also have many tasks to effectively manage patients living with heart failure, including ensuring that they are receiving timely and evidence-based care, coordinating with other members of the care team, and responding to acute patient issues like symptom worsening or the development of cardiac arrhythmias.
This description of the complexity and intensity of activities required for effective heart failure management helps illustrate how an effective digital and data infrastructure could significantly ease the management burden on both patients and care teams. A digital and data infrastructure could improve care coordination, identification of patients needing additional management, care efficiencies like reducing duplicative testing, and novel insights into disease phenotypes and new therapies. Specifically, digital health information systems such as remote patient monitoring, telehealth visits, EHRs, and digital messaging could allow for more seamless and rapid transmission of information between the patient and care team, facilitating coordinated, informed, and efficient care. The data from these information systems could also be analyzed to detect impending signs of symptom worsening, trigger patient and care team action items, and, ideally, prevent cardiac decompensation. This analysis could also help identify and alert the care team to any gaps in patient care. Coordinated digital health information systems could also address siloing of clinical information, allowing all care teams to access the entirety of the patient’s medical information and reducing inefficiencies that result from such fragmentation. Finally, data from large cohorts of heart failure patients could also be analyzed to identify novel phenotypes of the condition, suggest new hypotheses for their management, and facilitate pharmacological and device discoveries. The capabilities described above—and others—of a functional and interoperable digital and data ecosystem could significantly improve care experience, efficiency, and outcomes for patients living with heart failure and the experience clinicians have in delivering such care. The authors provide more detailed examinations of these potential benefits in the sections that follow.
Care Coordination
Care coordination reduces the potential for adverse health outcomes by organizing care activities across multiple providers and settings with the goal of safe, efficient, and quality care. Specifically, the American Heart Association and American College of Cardiology have advised that care coordination is crucial for patients with heart failure (Goldfarb et al., 2024). Studies show that heart failure patients with effective care coordination have lower morbidity, mortality, and rehospitalization rates and fewer adverse medication events (Samal et al., 2016). Costs associated with heart failure treatment in the US are estimated at $40 billion annually, including more than $10 billion due to emergent or unplanned care (Voigt et al., 2014).
Effective care coordination requires the exchange of current and comprehensive health information across a patient’s care team, regardless of health setting, medical discipline, or vendor health record platform. Unfortunately, studies indicate that nearly half of clinicians are unable to access patient information in real time from a different institution, forcing many to resort to manual, time-consuming methods to obtain these data (Anderson and Rowley, 2024). Seamless accessibility of clinically meaningful information requires a modern, adaptive digital and data architecture (National Academy of Medicine, 2018).
Health Monitoring and Communication
Recent examples of successful interoperable digital and data architecture that supports proactive self-management and improved outcomes for cardiac risk heart failure include wearable and remote monitoring devices, digital health tools, and health information networks and organizations. Personal health devices (e.g., smart watches or rings, wireless scales, remote oximetry) are increasingly used by care teams to help patients proactively avoid symptom exacerbation and manage heart failure symptoms (Sapna et al., 2023). These devices provide real time feedback to patients and providers on health status and needs and aim to reduce disease exacerbation and prevent health emergencies. They create real time data on physiological responses to current activities in the patient’s natural setting, offering in vivo feedback on behavior changes. When coupled with effective care management and patient education, patients often feel equipped to independently self-manage disease risk and respond to alerts, averting unnecessary adverse health outcomes.
Likewise, with proper networking and data quality assurance, these devices transmit clinical content that can integrate into patients’ EHRs, directly supporting care team workflow. When event notifications are integrated into the right workflow, the care team knows when to contact the patient on necessary medication or behavior changes to address cardiac concerns, ultimately reducing disease escalation and costly unplanned care (Lettere et al., 2016). For example, recent studies show that remote monitoring reduced heart failure hospital admission rates (Stevenson et al., 2023). Similarly, digital health interventions can leverage the power of connected care to improve heart failure outcomes. Tools like automated text messages, mobile health applications, telehealth, and decision support systems leverage the power of advanced computing and intelligence to synthesize the vast literature on heart failure treatment and present evidence-based suggestions within the provider workflow, quickly prompting provider actions and suggested outreach and management strategies for care teams (Myhre et al., 2024).
With fully interoperable health record platforms, patient data becomes readily available across different providers. Health information organizations are a network of interoperable systems allowing real time data exchange. The networks use a set of standards, services, and policies to securely exchange multiple data types from disparate sources in real time (Lettere et al., 2016). Given the foundational importance to effective care coordination of complex diseases like heart failure, health systems, industry leaders, and government agencies have invested in interoperability and data standards that promote health information exchange. Studies investigating the impact of health information networks on hospitals found that interoperability shortened hospital transfers by nearly 12 percent for heart attack patients (Li et al., 2022). Further, effective electronic communication and accessible health information also reduced 30-day readmissions in patients by nearly 3 percent (Li et al., 2022).
Effective and proactive self-management and care coordination for heart failure can have a significant positive impact on health outcomes and reduce the overall cost of care. While advances in digital health tools, personal health devices, and electronic information exchange show promise at reducing costly adverse events for patients with heart failure, work must continue to align data standards and technology nationally to avoid additional complications that risk patient safety or further fragment an already taxed medical ecosystem.
Precision Diagnostics and Management
Digital and data architecture initiatives that allow for more precision in identifying and treating patients with CVD have the potential to reduce costs. The application of precision medicine within CVD management is becoming more prevalent due to advancements in AI. The use of precision medicine is broadly classified into four domains: prediction, diagnosis, phenotyping, and risk stratification (Mohsen et al., 2023). Further stratified into early, mortality, disease, and dose, prediction applications comprise 50 percent of current precision medicine-focused studies (Mohsen et al., 2023). AI analytic techniques are a promising technology to support this goal. Specifically, AI-augmented CDS systems (AI-CDSS) can assist providers with identifying the most appropriate treatment options by applying ML algorithms to aggregated data from EHRs, imaging, laboratory results, and genomics databases (Bozyel et al., 2024). Capable of performing risk prediction, early detection, diagnosis, and treatment optimization, AI-CDSS can deliver cost saving benefits along the continuum of care (Bozyel et al., 2024). Below, the authors describe some early examples of using AI-CDSS to improve coronary artery disease detection, atrial fibrillation detection, and AI-assisted CVD phenotyping to support targeted therapies.
Coronary Artery Disease Detection
Risk stratification applications use ML to analyze Coronary Computed Tomography Angiography, where coronary inflammation, extent of coronary plaque, and other clinical factors are simultaneously assessed to quantify patient risk (Tsiachristas et al., 2024). Modeled over a patient’s lifetime, ML-enabled risk identification in clinical care led to a 4 percent reduction across myocardial infarctions, strokes, and heart failures while reducing cardiac mortality by 12 percent (Tsiachristas et al., 2024). With an estimated incremental cost-effectiveness ratio of $1,794 per quality-adjusted life year, ML-enabled risk modeling proves cost effective (Tsiachristas et al., 2024). AI technologies can also optimize the use of medical resources by automating portions of the diagnostic process while potentially improving prediction of coronary artery disease (CAD) progression and potential adverse events. AI-CDSS can prevent costly emergency interventions as well as reduce hospital readmission rates. AI-assisted monitoring within cardiovascular intensive care units allows for early identification of patients at risk for readmission, potentially saving millions in CAD-related inpatient costs per year when implemented at scale (Kessler et al., 2023; Moazemi et al., 2023).
Atrial Fibrillation Detection
AI is increasingly employed in novel ways that improve disease detection and discovery. In the area of CVD, AI-augmented electrocardiograms (EKGs) are being used for early detection of atrial fibrillation (AF). In a 2019 study by Attia et al., researchers leveraged a repository of over 400,000 EKGs performed at the Mayo Clinic to train the convolutional neural network (CNN) model. The AI-enabled EKG predicted AF with an overall accuracy of 83.3 percent even among asymptomatic patients (Attia et al., 2019). The incorporation of AI within EKGs can lead to improved patient outcomes and could likely lower costs associated with catastrophic and emergency care. As nearly one-third of AF patients are asymptomatic, early detection and intervention within this population could save nearly $8 billion of the $26 billion spent on treating AF patients annually (Reddy et al., 2024).
While standard EKG remains the most accepted method of AF diagnosis, the proliferation of photoplethysmography through the use of wearable devices creates an opportunity to include AI algorithms in the quest for advanced AF detection. Two separate studies conducted by Apple and Huawei both found that passive monitoring for AF using wearables is highly effective, achieving positive predictive values of 84 and 92 percent, respectively (Harmon et al., 2023). The advanced detection of AF, particularly in asymptomatic patients, enables early interventions such as anticoagulation therapy or ablation, lowering risk of stroke (Askarinejad et al., 2025).
AI-Assisted CVD Phenotyping
Despite the relative paucity of studies focused on AI-enabled phenotyping (14 percent), this practice represents perhaps one of the most consequential areas within precision cardiovascular medicine (Mohsen et al., 2023). Leveraging a combination of genomics and clinical data, AI-driven algorithms can be used to identify complex and chronic disease sub-phenotypes (Mohsen et al., 2023). In the case of AF, deep learning algorithms can be used to analyze potential responses to various treatments, identifying patients who may benefit from specific therapies like anticoagulants or catheter ablation (Krittanawong et al., 2017). These novel approaches to precision medicine could improve treatment efficacy and patient outcomes while simultaneously reducing adverse events and driving down cost.
The increased accessibility of generative AI tools is also empowering patients to engage in self-diagnostic discovery. Research and testimonials suggest that patients increasingly use large language models like ChatGPT to investigate unexplained symptoms, even in high-risk conditions like CVD (Stokel-Walker, 2024). Recent studies have sought to evaluate the accuracy of diagnosis-related information provided by these models. One specific study evaluated ChatGPT’s accuracy in providing recommendations on hypertension management, highlighting the potential of AI models to assist patients in understanding and managing cardiovascular conditions (Kassab et al., 2023). This study aligns with broader findings on patient-AI collaboration, where health consumers actively contribute to diagnostic refinement through AI-aided self-assessment—particularly when symptoms are subtle or intermittent, as is common in paroxysmal AF (Wu, 2024). Millenson has described this trend as emblematic of a shift toward “collaborative health”, where informed patients engage as partners in detection and decision making, aided by accessible AI models (Millenson, 2017).
Economic Impact
The lack of effective and interoperable digital and data infrastructure has profound economic consequences for health care. For example, poor visibility into care needs and a lack of coordinated care across settings and disciplines for heart failure patients often leads to duplicative procedures, unplanned high-cost care, and missed opportunities to prevent adverse outcomes. Expanding on the promise of interoperability to promote efficient health data exchange that improves quality, safety, and efficiency will also help provide high-quality care and reduce costs. While an effective and interoperable digital and data infrastructure necessitates strong governance that protects data privacy and standards to ensure interoperability across data types, the technology exists to support the deployment of such an architecture broadly. Specifically, advances in computing and the storage and development of predictive algorithms for Enterprise Master Patient Index systems support the combination of large and disparate data sets into a single clinical data repository that can better represent the many facets of care required for a single patient or patient population. Likewise, regulatory efforts on data standards as well as the incorporation of data classes for non-HIPAA protected data represent a promise to translate data across the entire ecosystem to health insights. As the learning capacity and computing power of AI grows, the use of precision cardiovascular medicine will likely proliferate. Integrated digital and data infrastructure has the potential to help realize significant cost savings across the health system. As the economic cost of care for heart failure in the US today soars, the value proposition for prioritizing interoperable, effective, and efficient data exchange is considerable.
Use Case 2: Maternal/Fetal Health and Maternal Mortality
Maternal Mortality and the Need for a Digital Health Infrastructure
In the US, maternal mortality rates—death during pregnancy or within 42 days after delivery—remain high at 18.6 deaths per 100,000 live births in 2023 (Hoyert, 2025). Black women experience a maternal mortality rate of 50.3 per 100,000 live births, more than two times higher than white women (Hoyert, 2025). Equally concerning are pregnancy-related deaths—which occur during pregnancy, delivery, or within a year after delivery. Significant disparities by race and ethnicity are noted in pregnancy-related deaths, with Black, American Indian and Alaska Native, and Pacific Islander women having the highest mortality rates (KFF, 2025b).
Understanding the underlying causes of pregnancy-related deaths and identifying opportunities for prevention are critical to improving fetal, infant, and maternal health outcomes. Prevention of pregnancy-related deaths and elimination of associated disparities will also help reduce severe maternal morbidity and avert adverse sequelae to the fetus and newborn—including stillbirth, infant mortality, preterm birth, and low birthweight (Zeitlin et al., 2019; Phibbs et al., 2022; Tsamantioti et al., 2025).
Addressing disparities in maternal mortality and pregnancy-related death requires a data infrastructure that identifies risks earlier than current systems can; ensures continuity of care throughout pregnancy, delivery, and postpartum; supports timely interventions; and identifies opportunities for improvement in quality of care. Currently, fragmented and inconsistent data systems hinder efforts to meet these needs, limiting health care providers’ and patients’ ability to make informed decisions based on comprehensive health information.
Fragmented Information
High quality maternal and infant care requires collaboration and data sharing between health care providers across multiple settings, including primary care, prenatal clinics, and hospitals—including emergency and pediatric care centers. However, data is often siloed in different systems, leading to gaps in care coordination (Rebecca A. Gourevitch, 2020; Chappel et al., 2021). For example, a mother’s prenatal care records at one facility may not be readily available to the team managing her delivery at another hospital, and maternal health and prenatal records may not be linked to the new infant’s record in the same hospital soon after delivery. This lack of integration results in incomplete health histories, missed risks, and delays in treatment during critical moments.
Health care systems also often collect and store information in varying formats, further complicating data sharing. A mother’s important preconception health information—such as lab results indicating prediabetes or blood pressure trends related to chronic hypertension—may not be accessible when she transfers care to another facility. Similarly, infant health records, including details on delivery complications or newborn screening results may not flow seamlessly between hospital systems and pediatric providers. These gaps in communication between systems contribute to fragmented care, which disproportionately affects underserved populations and exacerbates disparities in maternal and infant outcomes. Without full access to primary care and prenatal records, health care providers may be unaware of pregnancy-related complications or preexisting conditions that could influence decisions during labor and delivery.
Challenges with Standardization and Mother-Baby Data Linkage
Another issue is the lack of widely adopted standards for maternal and infant data. Without consistent and comparable data, it is difficult for health care providers to share crucial information across settings and address gaps in care. Key data elements, such as prenatal visit summaries, maternal health conditions, and newborn health metrics are not always standardized. This inconsistency leads to difficulties in linking records between different care settings, making it harder to provide seamless care.
Maternal and infant data are often stored in separate systems, creating additional challenges in linking information across health care providers. This disconnect is especially problematic during transitions of care, such as from prenatal to delivery or from hospital to home. Critical information is frequently lost in these transitions, preventing health care providers from having a full understanding of the patient’s history, which can lead to missed opportunities for early interventions. This gap also leads to difficulty in understanding potential linkages between maternal health and the emerging health of newborns over time.
Programs that aim to improve maternal and infant outcomes, such as Perinatal Quality Collaboratives and Alliance for Innovation in Maternal Health care bundles, have shown the benefits of coordinated care (American College of Obstetricians and Gynecologists, n.d.). However, these efforts are limited by the inconsistent integration of data across care sites. If a mother’s pregnancy complications or gestational diabetes history are not linked to the infant’s health records, pediatricians may lack essential information that could affect the management of the newborn’s health. This disconnect prevents providers from delivering fully informed, coordinated care. While techniques like Privacy Preserving Record Linkage, used in pediatric studies during the COVID-19 pandemic, offer models for improving data governance and security, such approaches are not widely implemented in maternal and infant care (National Institute of Child Health and Human Development, 2022). Without robust data linkage frameworks, the secure and accurate exchange of health information remains challenging, exacerbating the fragmentation of care and reducing the effectiveness of programs aimed at improving maternal and infant outcomes.
Access and Emerging Standards
There are, however, opportunities to accelerate interoperability under the 21st Century Cures Act. The 2023 updates to FHIR standards provide an example of how emerging data sharing frameworks can enhance access to care (HL7 International, 2022). FHIR enables smoother data exchange between health care systems, helping providers access maternal health metrics and infant records in real time. However, implementation challenges persist, particularly in maternity care systems where integration with legacy systems is often difficult.
In 2023, the Eunice Kennedy Shriver National Institute of Child Health and Human Development at the NIH worked with federal and private stakeholders to standardize the exchange of clinical maternal and infant health data, producing The Maternal & Infant Health Information for Research Implementation Guide. The Guide helps clinicians, academics, and others to capture, aggregate, and analyze data from different EHRs (HL7 International, 2022). Similarly, ASTP’s development of USCDI+ for maternal health aims to standardize a core set of data elements while supporting the collection of key information necessary for high-quality care and maternal health research (ASTP, 2025b). While these efforts are promising, full adoption and integration of these standards across urban and rural health care systems remain a challenge.
Artificial Intelligence and Machine Learning
AI and ML can assist clinical care by predicting risks, identifying complications early, and enabling real-time decision support.
Predictive Analytics
By analyzing large datasets, AI can help identify patterns and risk factors that might not be immediately apparent to clinicians, like preeclampsia, gestational diabetes, and preterm birth. For example, AI can combine data from prenatal visits, genetic information, and lifestyle factors to predict a mother’s risk of developing gestational diabetes. Early detection of these issues would allow for closer monitoring and timely interventions, which can prevent the condition from progressing and reduce the need for emergency interventions during labor.
Fetal Monitoring
Traditional fetal monitoring during labor relies heavily on manual interpretation of data from fetal heart rate monitors, which can lead to inconsistencies or missed warning signs. ML algorithms can analyze fetal heart rate patterns continuously, identifying subtle changes that might indicate distress, enabling earlier and more accurate interventions, reducing the risk of neonatal asphyxia, and improving birth outcomes overall. AI-based fetal monitoring systems are especially beneficial in high-risk pregnancies, where continuous, real-time analysis can make the difference between a straightforward and complicated delivery. Remote fetal monitoring has also demonstrated benefits in improving outcomes, particularly in high-risk pregnancies. A systematic review and metanalysis showed that remote fetal monitoring reduces the risk of neonatal asphyxia by 34 percent (Li et al., 2023). Remote monitoring tools allow for early detection of complications, providing valuable insights into maternal and fetal health during labor and helping reduce the need for emergency interventions (Mhajna et al., 2020; Martin et al., 2023).
Resource Allocation and Care Pathways
AI tools can also analyze hospital and clinic data to predict demand for labor and delivery services, helping health care administrators allocate resources more efficiently. These predictions can help ensure that labor and delivery units are properly equipped to handle both routine deliveries and emergencies. By anticipating demand, hospitals can also reduce wait times and improve overall patient experience.
CDS Systems (CDSS)
CDSS may alert a clinician when a patient’s vital signs or lab results indicate a potential complication, such as an impending hypertensive crisis. By providing actionable insights at the point of care, CDSS can improve patient safety and reduce the likelihood of adverse outcomes. However, broader population outcomes may be difficult to assess. In a systematic review of 684 studies by Lin and colleagues, the authors noted gaps in CDSS based on the need for additional maternal history, lack of community factors such as social determinants of health, and internal versus external validation (Lin et al., 2024).
Genomics
Clinicians could leverage whole-genome sequencing data to identify genetic markers associated with rare diseases that might affect the pregnancy or newborn. This is particularly important for families with a history of genetic disorders, recurrent pregnancy loss, or when a baby shows signs of developmental issues in utero. AI algorithms can help quickly sift through vast amounts of genetic data to identify mutations that may indicate a higher risk for conditions like congenital heart disease or other inherited disorders, allowing health care providers to counsel families on potential interventions or prepare for necessary care after birth (Clark et al., 2019).
Postpartum Care
Clinicians can predict the risk of postpartum hemorrhage or infection—two leading causes of maternal mortality—by analyzing patient data including delivery history, blood loss during labor, and vital signs (Venkatesh et al., 2020; Westcott et al., 2022; Lengerich et al., 2024). AI tools can help identify mothers who are at higher risk for these conditions and ensure that they receive closer monitoring and preventive care in the critical hours and days following delivery. In addition, new care opportunities exist with improved access to longer periods of postpartum care in 48 states, the District of Columbia, and United States Virgin Islands, all of which have expanded Medicaid coverage to one year after delivery (KFF, 2025a).
Health Care Disparities
AI tools can potentially inform interventions that improve access to care, ensure that all patients receive the same high level of attention and treatment regardless of demographic background, and reduce biases. However, AI can potentially exacerbate disparities if biases are integrated into algorithms. For example, when using vaginal birth after C-section (VBAC) calculators, Black women were systematically identified as less likely to successfully have a VBAC, leading to higher levels of Black women to be recommended for a C-section rather than attempting a VBAC (Vyas et al., 2019).
Automation of Administrative Tasks
AI can also help streamline tasks like appointment scheduling, billing, chart summarization, and patient triage, allowing health care providers to focus on patient care. For example, chatbots powered by natural language processing can help answer routine patient queries , schedule appointments, monitor for postpartum or infant health concerns, and provide personalized health advice based on the patient’s medical records (Nguyen et al., 2024; Rivera Rivera et al., 2024). In addition, providers could use predictive tools to analyze comprehensive datasets that span multiple care settings and identify potential complications earlier in pregnancy. These tools are more effective when they can draw data from a wide range of sources, improving the accuracy of risk assessments and enabling timely interventions.
Economic Impact
The lack of interoperable data systems in maternal health has significant economic implications. Adverse maternal health and associated morbidities are estimated to cost the health care system $32.3 billion from conception to 5 years postpartum (Paul, 2024). Currently, 25 percent of pregnant women do not receive adequate prenatal care (Martin and Osterman, 2023). The inability to access comprehensive patient records can lead to duplicative tests, unnecessary interventions, inaccurate diagnoses, and increased hospital admissions, driving up health care costs. For instance, preventing emergency C-sections and reducing neonatal ICU admissions through better data integration could save thousands of dollars per pregnancy (DeJoy et al., 2020). Complicating the interoperability of health data systems is the inability of surveillance programs to obtain data in a timely fashion. For example, SEER provides detailed cancer incidence and survival data from population-based cancer registries covering approximately 48 percent of the US population, but due to fragmented data systems and the need for high levels of curation, SEER data lags by two years (Murphy et al., 2024).
From a cost-benefit perspective, improving care coordination by implementing FHIR standards and adopting USCDI+ could reduce inefficiencies and improve outcomes. Studies suggest that reducing unnecessary interventions and improving access to necessary prenatal care through integrated data systems could result in savings of several hundred dollars per pregnancy, particularly through reductions in emergency procedures and hospital stays (Opiyo et al., 2020; Kuhlmann et al., 2021). Looking forward, implementing TEFCA could further support cross-system interoperability, enabling seamless data sharing and reducing administrative burdens.
The return on investment for implementing these data systems is significant. Health care systems that prioritize seamless data exchange can save on long-term care costs and reduce readmissions while improving maternal and infant health outcomes. Furthermore, the broader economic impact of improving maternal health extends beyond health care costs, as healthier mothers and infants contribute to increased productivity and societal well-being (Phibbs et al., 2020). AI and ML technologies also offer the potential to reduce health care costs by preventing complications and enabling more cost-effective interventions.
Use Case 3: Non–Small Cell Lung Cancer Novel Therapies
Checkpoint inhibitor therapy and targeted therapies have transformed the landscape of cancer treatment, particularly for non-small cell lung cancer (NSCLC) (de Jong et al., 2023). These therapies have shown remarkable promise, offering new hope to both newly diagnosed patients and those who have exhausted traditional treatments. Currently, over 40 percent of patients with newly diagnosed NSCLC possess targetable mutations, making them eligible for cutting-edge targeted therapies (Barlesi et al., 2016). However, the complexity and high cost of these therapies necessitate precise, personalized approaches to maximize their efficacy and accessibility. Digital health technologies have emerged as key facilitators in this domain, offering tools for personalized medicine, real-time monitoring of treatment responses, and data-driven optimization of therapeutic strategies.
NSCLC and the Need for a Digital Health Infrastructure
Despite advances and novel treatments, two-year survival in advanced NSCLC remains under 50 percent (Howlader et al., 2020). Therefore, personalized medicine and targeted therapies are not just a frontier of scientific innovation but a pressing public health need. Making effective treatments available is crucial for improving survival rates and quality of life for patients with cancer and other conditions. A major barrier to realizing this potential is access to appropriate molecular and genetic testing, which identifies patients who can benefit from targeted therapies. Unfortunately, over half of all patients with NSCLC do not receive comprehensive genomic profiling, with even greater disparities between Black and White individuals (Bruno et al., 2021). Furthermore, even among those with actionable mutations, 64 percent of eligible NSCLC patients do not receive targeted treatments, leading to increased negative health outcomes (Sadik et al., 2022; Scott et al., 2024).
Guideline Changes
Digital health technologies have the potential to revolutionize how NSCLC is treated by enabling more frequent automated updates to clinical guidelines, bypassing the delays associated with traditional updates such as those from the National Comprehensive Cancer Network. Using large language models, these updates could be produced more quickly, and, following appropriate final curation by medical experts, communicated and implemented into the workflow of nationwide health systems. A future where this is possible would require health system interoperability that permits rapid dissemination, providing physicians with details of the updated recommendations efficiently. Goals could be established, and progress towards them measured, by leveraging improved data flows and digital technologies that ingest and communicate this information.
Discovery
Advances in AI may also accelerate drug and biomarker discovery, paving the way for improved personalized treatment strategies. For example, automated biomarker identification using pathomics and radiomics has been shown to help identify novel biomarkers that can guide therapy selection (Madabhushi and Lee, 2016). Furthermore, leveraging EHRs can significantly improve cancer detection and the identification of specific genetic mutations. AI-driven multimodal predictors of immune checkpoint inhibitor benefit—which integrate data from histopathology, imaging, and genomics—can help stratify patients for optimal therapeutic responses (Artera, n.d.). The analysis of large genomic datasets using computational chemistry methods can also aid in identifying new drug targets and advancing the development of tailored treatments. Integration of multimodal data per patient aggregated across all patients with NSCLC could more rapidly improve the detection of co-mutations and other risk factors. These discoveries might increase or decrease treatment response rates and adverse experiences to refine new research and development efforts towards more safe and effective treatments. The lack of nationwide aggregation of such information limits the potential to accelerate learning and implement these learnings into new therapies.
Economic and Quality Impact
Remote monitoring using real-world datasets allows for continuous assessment of quality and safety metrics, providing a more dynamic and responsive approach to patient care (Beauchamp et al., 2020). For example, large real-world datasets may facilitate identifying clinically- and molecularly-defined subgroups in whom checkpoint inhibitor therapy is less effective, potentially reducing cost associated with ineffective treatments (Sun et al., 2021). Proactive adverse event monitoring facilitated by AI could also potentially enhance safety surveillance, informing regulatory bodies like the FDA for more timely intervention. EHR- and claims-based repositories offer the potential for real-time monitoring of targeted and novel therapies, ensuring that they are utilized when indicated. This approach would provide an advantage over current quality metrics, which often rely on self-reporting, delaying an accurate depiction of real-world practice. It would also support a more cost-effective health system by aligning treatments with those who are most likely to benefit and less likely to have adverse effects. By creating real-time systems that monitor guideline-recommended treatment approaches and providing feedback to physicians and health systems on their adherence, more patients could benefit from the rapid implementation of best practice guidelines (Sadik et al., 2022).
Digital health and AI can also play critical roles in improving surveillance, quality, prediction, and decision support in NSCLC treatment. Robust data systems are essential for monitoring treatment responses and adverse events, contributing to a more comprehensive understanding of patient outcomes. ML could aid in treatment optimization, reducing ineffective treatments, and avoiding unnecessary hospitalizations, ultimately leading to more appropriate and reduced spending. By leveraging digital health and AI, the health care ecosystem could move toward a more proactive, precise, and patient-centered approach to NSCLC care, unlocking the full potential of novel therapies and improving outcomes for patients. The integration of digital health technologies and AI into NSCLC treatment holds significant promise for improving access to novel therapies, enhancing survival and safety outcomes, and improving and reducing overall health care costs. Real-time monitoring, advanced data analytics, and personalized treatment approaches can help overcome existing barriers to effective NSCLC care, ultimately advancing the future of cancer therapy.
Use Case 4: Diabetes Mellitus
Diabetes mellitus is a critical global health challenge, affecting approximately 49 million Americans—about 16 percent of the population—with significant economic and health effects (CDC, 2024). The disease’s multisystem impact encompasses cardiovascular, renal, neurological, ophthalmologic, and metabolic complications, creating a complex clinical landscape that demands sophisticated management strategies (Bright and Sakurada, 2016). The economic burden of diabetes is also staggering. Direct medical costs and reduced productivity associated with diabetes amount to more than $413 billion annually in the US (CDC, 2024). Genetic predisposition plays a significant role, with first-degree relatives of individuals with type 2 diabetes experiencing a 2 to 6 times higher risk of developing the condition than their peers (Florez et al., 2003). Diabetes-related complications also represent a substantial population health challenge. Approximately 50 percent of adults with diabetes develop chronic kidney disease, 28 percent develop diabetic retinopathy, and 30 percent experience peripheral neuropathy (Bansal et al., 2014). The metabolic cascades triggered by uncontrolled diabetes also create complex interrelated health risks. Hyperglycemia initiates inflammatory processes that accelerate atherosclerosis, increase cardiovascular risk, and compromise multiple organ systems, which not only reduce patient quality of life but dramatically increase health care utilization and costs (Rezende et al., 2020).
Diabetes Mellitus and the Need for a Digital Health Infrastructure
Care Fragmentation
Diabetes management requires a multidisciplinary approach involving primary care physicians, endocrinologists, ophthalmologists, nephrologists, cardiologists, and specialized diabetes educators. The National Quality Forum (NQF) has established a comprehensive set of quality metrics for diabetes care, reflecting the increasing complexity of managing this chronic condition (NQF, 2015). NQF and other quality metrics have evolved significantly over the past decade, encompassing a holistic approach to diabetes management. The metrics now include glycemic control assessment (HbA1c monitoring), cardiovascular risk management, comprehensive complication screening, medication adherence tracking, patient self-management education, nutritional counseling, mental health screening, and comprehensive metabolic monitoring (NQF, 2015).
The pharmaceutical landscape for diabetes treatment has also undergone a remarkable transformation, with significant expansion in new treatment options that address the nuanced needs of different patient populations. These medications include GLP-1 receptor agonists, SGLT-2 inhibitors, and novel insulin formulations. These new medication classes have demonstrated significant advantages over traditional treatments, but also have substantial side effects. A meta-analysis of 11 clinical trials demonstrated a 12 percent reduction in major adverse cardiovascular events with these new medication classes, particularly GLP-1 receptor agonists and SGLT-2 inhibitors (Qiu et al., 2020).
The emergence of precision medicine has the potential to improve—but also further complicate—diabetes management. Pharmacogenomic studies now suggest that genetic profiling can predict individual patient responses to specific medication classes. A landmark study revealed that genetic variations can influence the efficacy of metformin by up to 35 percent, highlighting the need for increasingly personalized treatment approaches (Zhou et al., 2014). This increasing complexity necessitates advanced care coordination strategies. Patients with type 2 diabetes interact with an average of 4 health care specialists, over the course of their care creating significant challenges in maintaining a cohesive care plan if the specialists cannot access the same robust, interoperable data about the patient (Orozco-Beltrán et al., 2021).
Digital Health and Telemedicine
Digital health technologies have revolutionized diabetes management through sophisticated monitoring and intervention strategies. Telemedicine platforms have demonstrated remarkable efficacy in diabetes self-management, providing comprehensive, patient-centered care that extends beyond traditional clinical settings and has shown significant clinical outcomes. A systematic review of 107 clinical trials revealed that structured telehealth interventions achieved multiple key objectives among patients with diabetes, including:
- Reduced HbA1c levels by an average of 0.4-0.7 percent compared to standard care,
- Improved medication adherence rates,
- Enhanced patient education and self-management skills, and
- Decreased health care utilization through proactive monitoring (Lee et al., 2017).
Clinical implementation of these enhanced telehealth programs typically involves multimodal approaches that have demonstrated clinical efficacy, including:
- Video consultations with diabetes specialists,
- Remote monitoring of glucose data,
- Personalized digital care plans,
- Automated medication and lifestyle recommendations, and
- Real-time feedback and risk stratification.
Continuous glucose monitoring systems have emerged as a particularly transformative technology. One study demonstrated that continuous glucose monitoring reduced HbA1c levels by 0.5-1.0 percent compared to traditional methods (Martens et al., 2021). However, practical implementation of these systems still present significant challenges. Insurance coverage remains inconsistent, with many patients facing substantial out-of-pocket expenses. Technical barriers also persist, including device accuracy limitations, user interface complexity, and data interpretation challenges. Patient education is critical, as improper use can lead to misinterpretation of glucose trends and potentially inappropriate treatment decisions.
Diabetic Retinopathy Screening: Deep Learning Methodologies
Diabetic retinopathy is a complex complication that is identified through sophisticated screening methodologies. Deep learning algorithms have revolutionized early detection through advanced image analysis techniques. The core methodology involves CNNs trained on extensive retinal imaging datasets, which then process high-resolution fundus photographs, detecting microscopic vascular changes indicative of early-stage retinopathy.
The emergence of AI-assisted diabetic retinopathy screening has prompted significant updates in medical coding and reimbursement. The American Medical Association has introduced several new Current Procedural Terminology (CPT) codes specifically addressing AI-assisted retinal screening. Notably, CPT code 92229 was introduced in 2021 to describe computer-aided detection and diagnostic imaging interpretation of retinal images (Codify by AAPC, 2026). This code allows for separate billing of AI-assisted screening, providing a financial mechanism to support the adoption of advanced diagnostic technologies. CMS has also begun to recognize these AI-assisted screening methodologies, creating a reimbursement framework that encourages the integration of advanced diagnostic technologies in diabetes care. Currently, three FDA-approved AI-based diabetic retinopathy screening platforms exist, with studies showing a 37 percent reduction in missed diagnoses compared to traditional ophthalmological assessments (Rajesh et al., 2023).
Artificial Intelligence
AI-powered diagnostic tools are increasingly being used to enhance screening for diabetes complications, offering notable improvements in accuracy and efficiency. Deep learning algorithms have shown promise in detecting and predicting various diabetes-related health risks. For example, AI models for diabetic retinopathy screening have reached up to 95 percent accuracy in identifying early-stage retinal changes, which can help support timely interventions (Rajesh et al., 2023). In nephropathy prediction, ML models demonstrate 71 percent accuracy in forecasting kidney disease progression, providing insights that may guide preventive care (Makino et al., 2019). Similarly, AI algorithms for cardiovascular risk stratification show sensitivity exceeding 70 percent, offering another tool to help assess and manage risks associated with diabetes. These developments highlight the growing role of AI in supporting clinical decision making and improving care strategies for patients with diabetes.
Economic Impact
The potential economic benefits of incorporating advanced digital health technologies into diabetes management could be substantial (Sweet et al., 2020). Reductions in health care expenditures could stem from several factors, including preventive interventions that lower hospitalization rates, early AI-driven detection that helps avoid costly treatments for advanced complications, and continuous glucose monitoring that reduces the need for emergency care. A rigorous economic modeling study estimated that an advanced digital diabetes management program could lower health care costs by nearly $1,200 per participant within a year (Sweet et al., 2020). These findings underscore the potential for digital health tools to contribute to more cost-effective management of diabetes at both individual and population levels.
Join the Conversation
New from #NAMPerspectives: Toward a National Health Digital and Data Architecture. Read now: https://doi.org/10.31478/202603b
The health sector continues to lag in building the robust digital infrastructure needed to fully realize innovation. A new NAM discussion paper explores the path toward a health care digital and data architecture. https://doi.org/10.31478/202603b #NAMPerspectives

References
3GPP. n.d. About 3GPP. Available at: https://www.3gpp.org/about-us (accessed December 31, 2025).
Abernethy, A., L. Adams, M. Barrett, C. Bechtel, P. Brennan, A. Butte, J. Faulkner, E. Fontaine, S. Friedhoff, J. Halamka, M. Howell, K. Johnson, P. Lee, P. Long, D. McGraw, R. Miller, J. Perlin, D. Rucker, L. Sandy, L. Savage, L. Stump, P. Tang, E. Topol, R. Tuckson, and K. Valdes. 2022. The Promise of Digital Health: Then, Now, and the Future. NAM Perspectives. Discussion Paper, National Academy of Medicine, Washington, DC. https://doi.org/10.31478/202206e.
Adler-Milstein, J., and A. K. Jha. 2014. Health information exchange among U.S. hospitals: Who’s in, who’s out, and why? Healthcare 2(1):26–32. https://doi.org/10.1016/j.hjdsi.2013.12.005.
AIM (Alliance for Innovation on Maternal Health). n.d. HomeAvailable at: https://saferbirth.org/ (accessed December 31, 2025).
American Heart Association. 2024. What Is Cardiomyopathy? Available at: https://www.heart.org/en/health-topics/cardiomyopathy/what-is-cardiomyopathy-in-adults (accessed January 20, 2026).
American Lung Association. 2024. State of Lung Cancer: 2024 Report. Available at: https://www.lung.org/getmedia/12020193-7fb3-46b8-8d78-0e5d9cd8f93c/SOLC-2024.pdf (accessed December 31, 2025).
Anderson, J., and T. Rowley. 2024. Clinician needs still not being met. EHR Interoperability 2024. Available at: https://klasresearch.com/archcollaborative/report/ehr-interoperability-2024/604 (accessed December 31, 2025).
Arfaoui, G., P. Bisson, R. Blom, R. Borgaonkar, H. Englund, E. Félix, F. Klaedtke, P. K. Nakarmi, M. Näslund, P. O’Hanlon, J. Papay, J. Suomalainen, M. Surridge, J.-P. Wary, and A. Zahariev. 2018. A security architecture for 5G networks. IEEE Access 6:22466–22479. https://doi.org/10.1109/ACCESS.2018.2827419.
Arora, J., J. L. Mega, A. Abernethy, and W. Stadtlander III. 2022. Connecting real-world data to support public health efforts. NEJM Catalyst. https://catalyst.nejm.org/doi/full/10.1056/CAT.22.0040.
Artera. n.d. Our clinically available, laboratory-developed test. Available at: https://artera.ai/arteraai-prostate-cancer-test (accessed December 31, 2025).
Askarinejad, A, D. A. Lane, P. Sadeghipour, M. Haghjoo, and G. Y. H. Lip. 2025. Stroke prevention in atrial fibrillation: A narrative review of current evidence and emerging strategies. European Journal of Clinical Investigation 55:e70082. https://doi.org/10.1111/eci.70082.
ASTP (Assistant Secretary for Technology Policy). n.d. National trends in hospital and physician adoption of electronic health records. Available at: https://www.healthit.gov/data/quickstats/national-trends-hospital-and-physician-adoption-electronic-health-records (accessed January 1, 2026).
ASTP. 2025a. United States Core Data for Interoperability (USCDI). Available at: https://www.healthit.gov/isp/united-states-core-data-interoperability-uscdi (accessed January 6, 2026).
ASTP. 2025b. USCDI+. Available at: https://www.healthit.gov/topic/interoperability/uscdi-plus (accessed January 4, 2026).
ASTP/ONC (Assistant Secretary for Technology Policy/Office of the National Coordinator for Health IT). 2025. §170.315(b)(10) Electronic Health Information export. Available at: https://www.healthit.gov/test-method/electronic-health-information-export (accessed December 26, 2025).
ASTP/ONC HHS (Assistant Secretary for Technology Policy/Office of the National Coordinator for Health Information Technology, Department of Health and Human Services). 2024. “Health Data, Technology, and Interoperability: Trusted Exchange Framework and Common Agreement (TEFCA). Federal Register 89 FR 101722, December 16, 2024. Available at: https://www.federalregister.gov/documents/2024/12/16/2024-29163/health-data-technology-and-interoperability-trusted-exchange-framework-and-common-agreement-tefca (accessed December 31, 2025).
Attia, Z. I., P. A. Noseworthy, F. Lopez-Jimenez, S. J. Asirvatham, A. J. Deshmukh, B. J. Gersh, R. E. Carter, X. Yao, A. A. Rabinstein, B. J. Erickson, S. Kapa, and P. A. Friedman. 2019. An artificial intelligence-enabled ECG algorithm for the identification of patients with atrial fibrillation during sinus rhythm: A retrospective analysis of outcome prediction. The Lancet 394(10201):861–867. https://doi.org/10.1016/s0140-6736(19)31721-0.
Badr, J., A. Motulsky, and J. L. Denis. 2024. Digital health technologies and inequalities: A scoping review of potential impacts and policy recommendations. Health Policy 146:105122. https://doi.org/10.1016/j.healthpol.2024.105122.
Bansal, D., K. Gudala, H. Muthyala, H. P. Esam, R. Nayakallu, and A. Bhansali. 2014. Prevalence and risk factors of development of peripheral diabetic neuropathy in type 2 diabetes mellitus in a tertiary care setting. Journal of Diabetes Investigation 5(6):714–721. https://doi.org/10.1111/jdi.12223.
Barlesi, F., J. Mazieres, J.-P. Merlio, D. Debieuvre, J. Mosser, H. Lena, L. Ouafik, B. Besse, I. Rouquette, V. Westeel, F. Escande, I. Monnet, A. Lemoine, R. Veillon, H. Blons, C. Audigier-Valette, P.-P. Bringuier, R. Lamy, M. Beau-Faller, J.-L. Pujol, J.-C. Sabourin, F. Penault-Llorca, M .G. Denis, S. Lantuejoul, F. Morin, Q. Tran, P. Missy, A. Langlais, B. Milleron, J. Cadranel, J.-C. Soria, and G. Zalcman for the Biomarkers France contributors. 2016. Routine molecular profiling of patients with advanced non-small-cell lung cancer: Results of a 1-year nationwide programme of the French Cooperative Thoracic Intergroup (IFCT). The Lancet 387(10026): 1415–1426. https://doi.org/10.1016/s0140-6736(16)00004-0.
Beauchamp, U. L., H. Pappot, and C. Holländer-Mieritz. 2020. The use of wearables in clinical trials during cancer treatment: Systematic review. JMIR mHealth and uHealth 8(11):e22006. https://doi.org/10.2196/22006.
Benda, N. C., T. C. Veinot, C. J. Sieck, and J. S. Ancker. 2020. Broadband internet access is a social determinant of health! American Journal of Public Health 110(8):1123-1125. https://doi.org/10.2105/AJPH.2020.305784.
Blumenthal, D. 2011. Implementation of the federal health information technology initiative. New England Journal of Medicine 365(25):2426–2431. https://doi.org/10.1056/NEJMsr1112158.
Boonstra, A., A. Versluis, and J. F. J. Vos. 2014. Implementing electronic health records in hospitals: A systematic literature review. BMC Health Services Research 14:370. https://doi.org/10.1186/1472-6963-14-370.
Borges do Nascimento, I. J., H. Abdulazeem, L. T. Vasanthan, E. Z. Martinez, M. L. Zucoloto, L. Østengaard, N. Azzopardi-Muscat, T. Zapata, and D. Novillo-Ortiz. 2023. Barriers and facilitators to utilizing digital health technologies by healthcare professionals. npj Digital Medicine 6:161. https://doi.org/10.1038/s41746-023-00899-4.
Borrelli, E. P., P. Saad, N. Barnes and J. D. Lucaci. 2025. The influence of medication adherence on Medicare Star Ratings: A decade-long analysis of health plan performance. Journal of Managed Care & Specialty Pharmacy Vol. 31 Issue 5 Pages 512-519.
Boston Children’s Hospital. 2022. Multi-solving population data use with SMART® Bulk FHIR access. Available at: https://smarthealthit.org/multi-solving-population-data-use-with-smart-bulk-fhir-access/ (accessed January 1, 2026).
Boston Children’s Hospital. 2020. SMART. Available at: https://docs.smarthealthit.org/ (accessed December 26, 2025).
Boston Consulting Group. “E-commerce poised to capture 41% of global retail sales by 2027—Up from just 18% in 2017.” Press Release. October 31, 2023. Available at: https://www.bcg.com/press/31october2023-ecommerce-global-retail-sales (accessed January 1, 2026).
Bozyel, S., E. Şimşek, D. Koçyiğit Burunkaya, A. Güler, Y. Korkmaz, M. Şeker, M. Ertürk, and N. Keser. 2024. Artificial intelligence-based clinical decision support systems in cardiovascular diseases. The Anatolian Journal of Cardiology 28(2):74–86. https://doi.org/10.14744/AnatolJCardiol.2023.3685.
Bradley, C. J., K. R. Yabroff, B. Dahman, E. J. Feuer, A. Mariotto, and M. L. Brown. 2008. Productivity costs of cancer mortality in the United States: 2000–2020. Journal of the National Cancer Institute 100(24):1763–1770. https://doi.org/10.1093/jnci/djn384.
Bright, R. and B. Sakurada. 2016. A Population Health Strategy for Diabetes: New Partners, New Opportunities. NAM Perspectives. Discussion Paper, National Academy of Medicine, Washington, DC. https://doi.org/10.31478/201602d.
Bruno, D. S., L. M. Hess, X. Li, E. W. Su, Y. E. Zhu, and M. Patel. 2021. Racial disparities in biomarker testing and clinical trial enrollment in non-small cell lung cancer (NSCLC). Journal of Clinical Oncology 39(15_suppl):9005. https://doi.org/10.1200/JCO.2021.39.15_suppl.9005.
Cancer Research UK. 2025. Survival for lung cancer. Available at: https://www.cancerresearchuk.org/about-cancer/lung-cancer/survival (accessed January 3, 2026).
CareQuality. 2025. Home. Available at: https://carequality.org/ (accessed December 27, 2025).
CDC (Centers for Disease Control and Prevention). 2024. A report card: Diabetes in the United States infographic. Available at: https://www.cdc.gov/diabetes/communication-resources/diabetes-statistics.html (accessed January 3, 2026).
CMS (Centers for Medicare & Medicaid Services). 2025a. Health technology ecosystem. Available at: https://www.cms.gov/priorities/health-technology-ecosystem/overview (accessed December 27, 2025).
CMS. 2025b. The universal foundation. Available at: https://www.cms.gov/medicare/quality/cms-national-quality-strategy/universal-foundation (accessed December 29, 2025).
CMS. 2024. CMS Interoperability and Prior Authorization Final Rule (CMS-0057-F). Available at: https://www.cms.gov/cms-interoperability-and-prior-authorization-final-rule-cms-0057-f (accessed January 4, 2026).
CMS. 2020. CMS Interoperability and Patient Access Final Rule (CMS-9115-F). Available at: https://www.cms.gov/priorities/burden-reduction/overview/interoperability/policies-regulations/cms-interoperability-patient-access-final-rule-cms-9115-f (accessed January 4, 2026).
CERN (European Organization for Nuclear Research). n.d. The birth of the Web. Available at: https://www.home.cern/science/computing/birth-web (accessed January 4, 2026).
Chappel, A., N. DeLew, V. Grigorescu, and S. R. Smith. 2021. Addressing the maternal health crisis through improved data infrastructure: Guiding principles for progress. Health Affairs Forefront. https://doi.org/10.1377/forefront.20210729.265068.
Charles, D., M. Gabriel, and M. F. Furukawa. 2014. Adoption of electronic health record systems among U.S. non-federal acute care hospitals: 2008–2013. ONC Data Brief 16. Available at: https://www.healthit.gov/sites/default/files/oncdatabrief16.pdf (accessed January 4, 2026).
Chow, C. K., J. Redfern, G. S. Hillis, J. Thakkar, K. Santo, M. L. Hackett, S. Jan, N. Graves, L. de Keizer, T. Barry, S. Bompoint, S. Stepien, R. Whittaker, A. Rodgers, and A. Thiagalingam. 2015. Effect of lifestyle-focused text messaging on risk factor modification in patients with coronary heart disease: A randomized clinical trial. JAMA 314(12):1255–1263. https://doi.org/10.1001/jama.2015.10945.
Christou, E., and F. Piller. 2024. “Organizational transformation: A management research perspective.” In Transformation towards sustainability: A novel interdisciplinary framework from rwth aachen university, edited by Peter Letmathe, Christine Roll, Almut Balleer, Stefan Böschen, Wolfgang Breuer, Agnes Förster, Gabriele Gramelsberger, Kathrin Greiff, Roger Häußling, Max Lemme, Michael Leuchner, Maren Paegert, Frank T. Piller, Elke Seefried and Thorsten Wahlbrink, 303-330. Cham: Springer International Publishing.
Cisco Systems. 2023. Understand and use the enhanced interior gateway routing protocol. Available at: https://www.cisco.com/c/en/us/support/docs/ip/enhanced-interior-gateway-routing-protocol-eigrp/16406-eigrp-toc.html (accessed January 5, 2026).
Clark, M. M., A. Hildreth, S. Batalov, Y. Ding, S. Chowdhury, K. Watkins, K. Ellsworth, B. Camp, C. I. Kint, C. Yacoubian, L. Farnaes, M. N. Bainbridge, C. Beebe, J. J. A. Braun, M. Bray, J. Carroll, J. A. Cakici, S. A. Caylor, C. Clarke, M. P. Creed, J. Friedman, A. Frith, R. Gain, M. Gaughran, S. George, S. Gilmer, J. Gleeson, J. Gore, H. Grunenwald, R. L. Hovey, M. L. Janes, K. Lin, P. D. McDonagh, K. McBride, P. Mulrooney, S. Nahas, D. Oh, A. Oriol, L. Puckett, Z. Rady, M. G. Reese, J. Ryu, L. Salz, E. Sanford, L. Stewart, N. Sweeney, M. Tokita, L. Van Der Kraan, S. White, K. Wigby, B. Williams, T. Wong, M. S. Wright, C. Yamada, P. Schols, J. Reynders, K. Hall, D. Dimmock, N. Veeraraghavan, T. Defay, and S. F. Kingsmore. 2019. Diagnosis of genetic diseases in seriously ill children by rapid whole-genome sequencing and automated phenotyping and interpretation. Science Translational Medicine 11(489). https://doi.org/10.1126/scitranslmed.aat6177.
Cleland, J. G. F., A. A. Louis, A. S. Rigby, U. Janssens, and A. H. M. M. Balk, and the TEN-HMS Investigators. 2005. Noninvasive home telemonitoring for patients with heart failure at high risk of recurrent admission and death: The Trans-European Network-Home-Care Management System (TEN-HMS) study. Journal of the American College of Cardiology 45(10):1654–1664. https://doi.org/10.1016/j.jacc.2005.01.050.
Codify by AAPC. 2026. CPT® 92229, Under ophthalmoscopy procedures. Available at: https://www.aapc.com/codes/cpt-codes/92229?srsltid=AfmBOorI0iNpcV6EPOkXu4UR3fgGBEUrCissWXqMx19IGZtzXBzAGM5z (accessed January 4, 2026).
CommonWell Health Alliance. 2025. Home. Available at: https://www.commonwellalliance.org/ (accessed December 27, 2025).
David, E. A., M. E. Daly, C.-S. Li, C.-L. Chiu, D. T. Cooke, L. M. Brown, J. Melnikow, K. Kelly, and R. J. Canter. 2017. Increasing rates of no treatment in advanced-stage non-small cell lung cancer patients: A propensity-matched analysis. Journal of Thoracic Oncology 12(3):437–445. https://doi.org/10.1016/j.jtho.2016.11.2221.
Dayer, L., S. Heldenbrand, P. Anderson, P. O. Gubbins, and B. C. Martin. 2013. Smartphone medication adherence apps: Potential benefits to patients and providers. Journal of the American Pharmacists Association 53(2):172–181. https://doi.org/10.1331/JAPhA.2013.12202.
de Jong, D., J. Das, H. Ma, J. P. Valiplackal, C. Prendergast, T. Roa, B. Braumuller, A. Deng, L. Dercle, R. Yeh, M. M. Salvatore, and K. M. Capaccione. 2023. Novel targets, novel treatments: The changing landscape of non-small cell lung cancer. Cancers 15(10):2855. https://doi.org/10.3390/cancers15102855.
DeJoy, S. A., M. G. Bohl, K. Mahoney, and C. Blake. 2020. Estimating the financial impact of reducing primary cesareans. Journal of Midwifery & Women’s Health 65(1):56-63. https://doi.org/10.1111/jmwh.13010.
Diamond, J., and A. D. DeVore. 2022. New strategies to prevent rehospitalizations for heart failure. Current Treatment Options in Cardiovascular Medicine 24(12):199–212. https://doi.org/10.1007/s11936-022-00969-y.
DuBard, C. A., W. H. Shrank, S. Cavanaugh, and F. Mostashari. 2024. Why the Star Ratings medication adherence measures must go. Health Affairs Forefront. https://doi.org/10.1377/forefront.20240103.310349.
Dutkiewicz, E., X. Costa-Perez, I. Z. Kovacs, and M. Mueck. 2017. Massive machine-type communications. IEEE Network 31(6):6–7. https://doi.org/10.1109/MNET.2017.8120237.
EHR Good Neighbor. n.d. BulkUP – the Bulk Data User Partnership. Available at: https://good-neighbor.smarthealthit.org/performance/ (accessed January 4, 2026).
Epic. 2025. Care Everywhere. Available at: https://www.epic.com/careeverywhere/?search=&country=&usstate= (accessed December 27, 2025).
Everson, J., J. C. Rubin, and C. P. Friedman. 2020. Reconsidering hospital EHR adoption at the dawn of HITECH: Implications of the reported 9% adoption of a “basic” EHR. Journal of the American Medical Informatics Association 27(8):1198–1205. https://doi.org/10.1093/jamia/ocaa090.
FCC (Federal Communications Commission). 2022. Advancing broadband connectivity as a social determinant of health. Available at: https://www.fcc.gov/health/SDOH (accessed January 4, 2026).
FDA (U.S. Food & Drug Administration). 2020. What is digital health? Available at: https://www.fda.gov/medical-devices/digital-health-center-excellence/what-digital-health (accessed January 4, 2026).
FINRA (Financial Industry Regulatory Authorty). n.d. About FINRA. Available at: https://www.finra.org/about (accessed January 5, 2026).
Florez, J. C., J. Hirschhorn, and D. Altshuler. 2003. The inherited basis of diabetes mellitus: Implications for the genetic analysis of complex traits. Annual Review of Genomics and Human Genetics 4:257–291. https://doi.org/10.1146/annurev.genom.4.070802.110436.
Galea, S., and M. Abba-Aji. 2024. New directions for population health science and scholarship. JAMA Health Forum 5(11):e244566. https://doi.org/10.1001/jamahealthforum.2024.4566.
Garcia, R. A., J. A. Spertus, M. C. Benton, P. G. Jones, D. B. Mark, J. D. Newman, S. Bangalore, W. E. Boden, G. W. Stone, H. R. Reynolds, J. S. Hochman, D. J. Maron, and the ISCHEMIA Research Group. 2022. Association of medication adherence with health outcomes in the ISCHEMIA trial. Journal of the American College of Cardiology 80(8):755–765. https://doi.org/10.1016/j.jacc.2022.05.045.
Garrido, M. M., H. G. Prigerson, Y. Bao, and P. K. Maciejewski. 2016. Chemotherapy use in the months before death and estimated costs of care in the last week of life. Journal of Pain and Symptom Management 51(5):875–881. https://doi.org/10.1016/j.jpainsymman.2015.12.323.
Gentili, A., G. Failla, A. Melnyk, V. Puleo, G. L. Di Tanna, W. Ricciardi, and F. Cascini. 2022. The cost-effectiveness of digital health interventions: A systematic review of the literature. Frontiers in Public Health 10. https://doi.org/10.3389/fpubh.2022.787135.
Gildea, T. R., S. DaCosta Byfield, D. K. Hogarth, D. S. Wilson, and C. C. Quinn. 2017. A retrospective analysis of delays in the diagnosis of lung cancer and associated costs. Clinicoeconmics and Outcomes Research 9:261–269. https://doi.org/10.2147/ceor.S132259.
Goldfarb, M. J., M. Abshire Saylor, B. Bozkurt, J. Code, K. E. Di Palo, A. Durante, K. Flanary, R. Masterson Creber, M. O. Ogunniyi, F. Rodriguez, and M. Gulati, on behalf of the American Heart Association Council on Clinical Cardiology, Council on Cardiovascular and Stroke Nursing, Council on Hypertension, Council on Lifestyle and Cardiometabolic Health, Council on Peripheral Vascular Disease, and Council on Quality of Care and Outcomes Research. 2024. Patient-centered adult cardiovascular care: A scientific statement from the American Heart Association. Circulation 149(20):e1176–e1188. https://doi.org/10.1161/cir.0000000000001233.
Gourevitch, R. A., A. F. Peahl, M. McConnell, and N. Shah. 2020. Understanding the impact of prenatal care: Improving metrics, data, and evaluation. Health Affairs Forefront. https://doi.org/10.1377/forefront.20200221.833522.
Guzman, C. E. V., and J. Heintzman. 2024. Expanding participatory data analysis: An innovative strategy for engaging Community Advisory Board (CAB) in qualitative research data analysis virtually. Journal of Participatory Research Methods 5(4):44–55. https://doi.org/10.35844/001c.126305.
Gwira, J. A., C. D. Fryar, and Q. Gu. 2024. Prevalence of total, diagnosed, and undiagnosed diabetes in adults: United States, August 2021–August 2023. NCHS Data Brief No. 516. Available at: https://www.cdc.gov/nchs/data/databriefs/db516.pdf (accessed January 3, 2026).
Hafeez, M., P. Achar, M. Neeralagi, and G. T. Naik. 2022. Correlation between diabetic retinopathy and diabetic peripheral neuropathy in patients with Type II diabetes mellitus. Journal of Pharmacy & Bioallied Sciences 14(Suppl 1):s658–s661. https://doi.org/10.4103/jpbs.jpbs_138_22.
Harmon, D. M., O. Sehrawat, M. Maanja, J. Wight, and P. Noseworthy. 2023. Artificial intelligence for the detection and treatment of atrial fibrillation. Arrhythmia and Electrophysiology Review 12:e12. https://doi.org/10.15420/aer.2022.31.
HIMSS (Healthcare Information and Management Systems Society). n.d. Interoperability in healthcare. Available at: https://nationalcapitalarea.himss.org/resources/interoperability-healthcare (accessed January 20, 2026).
HL7 International. 2025. CDS Hooks. Available at: https://cds-hooks.hl7.org/ (accessed December 26, 2025).
HL7 International. 2022. Longitudinal maternal & infant health information for research. Available at: https://hl7.org/fhir/us/mihr/index.html (accessed January 4, 2026).
Howell, M. D. 2024. Generative artificial intelligence, patient safety and healthcare quality: A review. BMJ Quality & Safety 33(11):748–754. https://doi.org/10.1136/bmjqs-2023-016690.
Howlader, N., G. Forjaz, M. J. Mooradian, R. Meza, C. Y. Kong, K. A. Cronin, A. B. Mariotto, D. R. Lowy, and E. J. Feuer. 2020. The effect of advances in lung-cancer treatment on population mortality. New England Journal of Medicine 383(7):640–649. https://doi.org/10.1056/NEJMoa1916623.
Hoyert, D. 2025. Maternal mortality rates in the United States, 2023. NCHS Health E-Stats. https://doi.org/https://dx.doi.org/10.15620/cdc/174577.
IEEE (Institute of Electrical and Electronics Engineers). n.d. IEEE: Advancing technology for humanity. Available at: https://www.ieee.org/ (accessed January 5, 2026).
ISO (International Organization for Standardization). n.d. ISO: Global standards for trusted goods and services. Available at: https://www.iso.org/home.html (accessed January 5, 2026).
ITU (International Telecommunication Union). n.d. About The International Telecommunication Union (ITU). Available at: https://www.itu.int/en/about/Pages/default.aspx (accessed January 5, 2026).
ITU. 2022. Global connectivity report 2022. Available at: https://www.itu.int/itu-d/reports/statistics/global-connectivity-report-2022/ (accessed January 4, 2026).
Jones, J. R., D. Gottlieb, A. J. McMurry, A. Atreja, P. M. Desai, B. E. Dixon, P. R. O. Payne, A. J. Saldanha, P. Shankar, Y. Solad, A. B. Wilcox, M. S. Ali, E. Kang, A. M. Martin, E. Sprouse, D. E. Taylor, M. Terry, V. Ignatov, K. D. Mandl, and the SMART Cumulus Network. 2024. Real world performance of the 21st Century Cures Act population-level application programming interface. Journal of the American Medical Informatics Association 31(5):1144–1150. https://doi.org/10.1093/jamia/ocae040.
Juhn, E., and E. Galvez. 2022. Incentivizing data sharing among health plans, hospitals, and providers to improve quality. The American Journal of Managed Care 28(12):e426–e427. https://doi.org/10.37765/ajmc.2022.89277.
Kassab, J., J. El Dahdah, M. Chedid El Helou, H. Layoun, A. Sarraju, L. J. Laffin, L. Cho, S. R. Kapadia, P. Collier, and S. C. Harb. 2023. Assessing the accuracy of an online chat-based artificial intelligence model in providing recommendations on hypertension management in accordance with the 2017 American College of Cardiology/American Heart Association and 2018 European Society of Cardiology/European Society of Hypertension guidelines. Hypertension 80(7):e125-e127. https://doi.org/10.1161/hypertensionaha.123.21183.
Kessler, S., D. Schroeder, S. Korlakov, V. Hettlich, S. Kalkhoff, S. Moazemi, A. Lichtenberg, F. Schmid, and H. Aubin. 2023. Predicting readmission to the cardiovascular intensive care unit using recurrent neural networks. Digital Health 9. https://doi.org/10.1177/20552076221149529.
KFF. 2025a. Medicaid postpartum coverage extension tracker. Available at: https://www.kff.org/medicaid/medicaid-postpartum-coverage-extension-tracker/ (accessed January 4, 2025).
KFF. 2025b. Racial disparities in maternal and infant health: Current status and key issues. Available at: https://www.kff.org/racial-equity-and-health-policy/racial-disparities-in-maternal-and-infant-health-current-status-and-key-issues/ (accessed January 21, 2026).
Krittanawong, C., H. Zhang, Z. Wang, M. Aydar, and T. Kitai. 2017. Artificial intelligence in precision cardiovascular medicine. Journal of the American College of Cardiology 69(21):2657–2664. https://doi.org/10.1016/j.jacc.2017.03.571.
Kuhlmann, P. K., D. N. Patel, A. Chen, J. Houman, J. Weinberger, L. N. Wood Thum, J. T. Anger, and K. S. Eilber. 2021. Economic evaluation of elective cesarean versus vaginal delivery on cost of future pelvic floor disorders in the United States. Neurourology and Urodynamics 40(1):451–460. https://doi.org/10.1002/nau.24582.
Kumar, A., A. Chhangani, J. Lassiter, and K. Haar. 2024. Standards and interoperability: The future of the global financial system. Atlantic Council. April 10, 2024. Available at: https://www.atlanticcouncil.org/in-depth-research-reports/issue-brief/standards-and-interoperability-the-future-of-the-global-financial-system/ (accessed January 5, 2026).
Lamarre, E., S. Chheda, M. Riba, V. Genest, and A. Nizam. 2023. The value of digital transformation. Harvard Business Review. July 31, 2023. Available at: https://hbr.org/2023/07/the-value-of-digital-transformation (accessed January 5, 2026).
Lee, S. W. H., C. K. Y. Chan, S. S. Chua, and N. Chaiyakunapruk. 2017. Comparative effectiveness of telemedicine strategies on type 2 diabetes management: A systematic review and network meta-analysis. Scientific Reports 7:12680. https://doi.org/10.1038/s41598-017-12987-z.
Lengerich, B. J., R. Caruana, I. Painter, W. B. Weeks, K. Sitcov, and V. Souter. 2024. Interpretable machine learning predicts postpartum hemorrhage with severe maternal morbidity in a lower-risk laboring obstetric population. American Journal of Obstetrics & Gynecology Maternal and Fetal Medicine 6(8):101391. https://doi.org/10.1016/j.ajogmf.2024.101391.
Lettere, M., D. Guerri, S. La Manna, M. C. Groccia, D. Lofaro, and D. Conforti. 2016. HeartDrive: A broader concept of interoperability to implement care processes for heart failure. Studies in Health Technology and Informatics 224:158–163. https://doi.org/10.3233/978-1-61499-653-8-158.
Li, Y., L. X. Lu, S. F. Lu, and J. Chen. 2022. The value of health information technology interoperability: Evidence from interhospital transfer of heart attack patients. Manufacturing & Service Operations Management 24(2):827–845. https://doi.org/10.1287/msom.2021.1007.
Li, S., Q. Yang, S. Niu, and Y. Liu. 2023. Effectiveness of remote fetal monitoring on maternal-fetal outcomes: Systematic review and meta-analysis. JMIR mHealth and uHealth 11:e41508. https://doi.org/10.2196/41508.
Lin, X., C. Liang, J. Liu, T. Lyu, N. Ghumman, and B. Campbell. 2024. Artificial intelligence-augmented clinical decision support systems for pregnancy care: Systematic review. Journal of Medical Internet Research 26:e54737. https://doi.org/10.2196/54737.
Little, R. J., R. D’Agostino, M. L. Cohen, K. Dickersin, S. S. Emerson, J. T. Farrar, C. Frangakis, J. W. Hogan, G. Molenberghs, S.A. Murphy, J. D. Neaton, A. Rotnitzky, D. Scharfstein, W. J. Shih, J. P. Siegel, and H. Stern. 2012. The prevention and treatment of missing data in clinical trials. New England Journal of Medicine 367:1355–1360. https://doi.org/10.1056/NEJMsr1203730.
Madabhushi, A., and G. Lee. 2016. Image analysis and machine learning in digital pathology: Challenges and opportunities. Medical Image Analysis 33:170–175. https://doi.org/10.1016/j.media.2016.06.037.
Maddox, T. M., J. L. Januzzi, Jr., L. A. Allen, K. Breathett, S. Brouse, J. Butler, L. L. Davis, G. C. Fonarow, N. E. Ibrahim, J. Lindenfeld, F. A. Masoudi, S. R. Motiwala, E. Oliveros, M. N. Walsh, A. Wasserman, C. W. Yancy, and Q. R. Youmans. 2024. 2024 ACC expert consensus decision pathway for treatment of heart failure with reduced ejection fraction: A report of the American College of Cardiology Solution Set Oversight Committee. Journal of the American College of Cardiology 83(15):1444–1488. https://doi.org/10.1016/j.jacc.2023.12.024.
Makino, M., R. Yoshimoto, M. Ono, T. Itoko, T. Katsuki, A. Koseki, M. Kudo, K. Haida, J. Kuroda, R. Yanagiya, E. Saitoh, K. Hoshinaga, Y. Yuzawa, and A. Suzuki. 2019. Artificial intelligence predicts the progression of diabetic kidney disease using big data machine learning. Scientific Reports 9:11862. https://doi.org/10.1038/s41598-019-48263-5.
Maliha, G., S. Gerke, I. G. Cohen, and R. B. Parikh. 2021. Artificial intelligence and liability in medicine: Balancing safety and innovation. The Milbank Quarterly 99(3):629–647. https://doi.org/10.1111/1468-0009.12504.
Mandel, J. C., D. A. Kreda, K. D. Mandl, I. S. Kohane, and R. B. Ramoni. 2016. SMART on FHIR: A standards-based, interoperable apps platform for electronic health records. Journal of the American Medical Informatics Association 23(5):899–908. https://doi.org/10.1093/jamia/ocv189.
Mandl, K. D. 2025. How AI could reshape health care—Rise in direct-to-consumer models. JAMA 333(19):1667–1669. https://doi.org/10.1001/jama.2025.0946.
Mandl, K. D., and I. S. Kohane. 2020. Data citizenship under the 21st Century Cures Act. New England Journal of Medicine 382(19):1781–1783. https://doi.org/10.1056/NEJMp1917640.
Mandl, K. D., and I. S. Kohane. 2017. A 21st-century health IT system—Creating a real-world information economy. New England Journal of Medicine 376(20):1905–1907. https://doi.org/10.1056/NEJMp1700235.
Mandl, K. D., and I. S. Kohane. 2016. Time for a patient-driven health information economy? New England Journal of Medicine 374(3):205–208. https://doi.org/10.1056/NEJMp1512142.
Mandl, K. D., and I. S. Kohane. 2009. No small change for the health information economy. New England Journal of Medicine 360(13):1278–1281. https://doi.org/10.1056/NEJMp0900411.
Mandl, K. D., and E. D. Perakslis. 2021. HIPAA and the leak of “deidentified” EHR data. New England Journal of Medicine 384(23):2171–2173. https://doi.org/10.1056/NEJMp2102616.
Mandl, K. D., D. Gottlieb, J. C. Mandel, V. Ignatov, R. Sayeed, G. Grieve, J. Jones, A. Ellis, and A. Culbertson. 2020. Push button population health: The SMART/HL7 FHIR bulk data access application programming interface. npj Digital Medicine 3:151. https://doi.org/10.1038/s41746-020-00358-4.
Martens, T., R. W. Beck, R. Bailey, K. J. Ruedy, P. Calhoun, A. L. Peters, R. Pop-Busui, A. Philis-Tsimikas, S. Bao, G. Umpierrez, G. Davis, D. Kruger, A. Bhargava, L. Young, J. B. McGill, G. Aleppo, Q. T. Nguyen, I. Orozco, W. Biggs, K. J. Lucas, W. H. Polonsky, J. B. Buse, D. Price, and R. M. Bergenstal, for the MOBILE Study Group. 2021. Effect of continuous glucose monitoring on glycemic control in patients with type 2 diabetes treated with basal insulin: A randomized clinical trial. JAMA 325(22):2262–2272. https://doi.org/10.1001/jama.2021.7444.
Martin, J. A., and M. J. K. Osterman. 2023. Changes in prenatal care utilization: United States, 2019–2021. National Vital Statistics Reports 72(4):1–14. Available at: https://www.cdc.gov/nchs//data/nvsr/nvsr72/nvsr72-04.pdf (accessed January 5, 2026).
Martin, J. K., E. G. Price-Haywood, M. M. Gastanaduy, D. G. Fort, M. K. Ford, S. P. Peterson, and J. R. Biggio. 2023. Unexpected term neonatal intensive care unit admissions and a potential role for centralized remote fetal monitoring. American Journal of Perinatology 40(3):297–304. https://doi.org/10.1055/s-0041-1727214.
Martin, S. S., A. W. Aday, Z. I. Almarzooq, C. A. M. Anderson, P. Arora, C. L. Avery, C. M. Baker-Smith, B. Barone Gibbs, A. Z. Beaton, A. K. Boehme, Y. Commodore-Mensah, M. E. Currie, M. S. V. Elkind, K. R. Evenson, G. Generoso, D. G. Heard, S. Hiremath, M. C. Johansen, R. Kalani, D. S. Kazi, D. Ko, J. Liu, J. W. Magnani, E. D. Michos, M. E. Mussolino, S. D. Navaneethan, N. I. Parikh, S. M. Perman, R. Poudel, M. Rezk-Hanna, G. A. Roth, N. S. Shah, M.-P. St-Onge, E. L. Thacker, C. W. Tsao, S. M. Urbut, H. G. C. Van Spall, J. H. Voeks, N.-Y. Wang, N. D. Wong, S. S. Wong, K. Yaffe, and L. P. Palaniappan, on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. 2024. 2024 heart disease and stroke statistics: A report of US and global data from the American Heart Association. Circulation 149(8):e347–e913. https://doi.org/10.1161/cir.0000000000001209.
McGinnis, J. M., H. V. Fineberg, and V. J. Dzau. Shared commitments for health and health care: A trust framework from the learning health system. NAM Perspectives. Commentary, National Academy of Medicine, Washington, DC. https://doi.org/10.31478/202412c.
McGraw, D., and K. D. Mandl. 2021. Privacy protections to encourage use of health-relevant digital data in a learning health system. npj Digital Medicine 4:2. https://doi.org/10.1038/s41746-020-00362-8.
McKinney, S. M., M. Sieniek, V. Godbole, J. Godwin, N. Antropova, H. Ashrafian, T. Back, M. Chesus, G. S. Corrado, A. Darzi, M. Etemadi, F. Garcia-Vicente, F. J. Gilbert, M. Halling-Brown, D. Hassabis, S. Jansen, A. Karthikesalingam, C. J. Kelly, D. King, J. R. Ledsam, D. Melnick, H. Mostofi, L. Peng, J. J. Reicher, B. Romera-Paredes, R. Sidebottom, M. Suleyman, D. Tse, K. C. Young, J. De Fauw, and S. Shetty. 2020. International evaluation of an AI system for breast cancer screening. Nature 577(7788):89–94. https://doi.org/10.1038/s41586-019-1799-6.
McMurry, A. J., D. I. Gottlieb, T. A. Miller, J. R. Jones, A. Atreja, J. Crago, P. M. Desai, B. E. Dixon, M. Garber, V. Ignatov, L. A. Kirchner, P. R. O. Payne, A. J. Saldanha, P. R. V. Shankar, Y. V. Solad, E. A. Sprouse, M. Terry, A. B. Wilcox, and K. D. Mandl. 2024. Cumulus: A federated electronic health record-based learning system powered by Fast Healthcare Interoperability Resources and artificial intelligence. Journal of the American Medical Informatics Association 31(8):1638–1647. https://doi.org/10.1093/jamia/ocae130.
Mhajna, M., N. Schwartz, L. Levit-Rosen, S. Warsof, M. Lipschuetz, M. Jakobs, J. Rychik, C. Sohn, and S. Yagel. 2020. Wireless, remote solution for home fetal and maternal heart rate monitoring. American Journal of Obstetrics & Gynecology Maternal and Fetal Medicine 2(2):100101. https://doi.org/10.1016/j.ajogmf.2020.100101.
Millenson, M. L. 2000. Demanding Medical Excellence: Doctors and Accountability in the Information Age. University of Chicago Press: Chicago, IL.
Millenson, M. L. 2017. When “patient centred” is no longer enough: The challenge of collaborative health: An essay by Michael L Millenson. BMJ 358:j3048. https://doi.org/10.1136/bmj.j3048.
Moazemi, S., S. Vahdati, J. Li, S. Kalkhoff, L. J. V. Castano, B. Dewitz, R. Bibo, P. Sabouniaghdam, M. S. Tootooni, R. A. Bundschuh, A. Lichtenberg, H. Aubin, and F. Schmid. 2023. Artificial intelligence for clinical decision support for monitoring patients in cardiovascular ICUs: A systematic review. Frontiers in Medicine 10. https://doi.org/10.3389/fmed.2023.1109411.
Mohsen, F., B. Al-Saadi, N. Abdi, S. Khan, and Z. Shah. 2023. Artificial intelligence-based methods for precision cardiovascular medicine. Journal of Personalized Medicine 13(8):1268. https://doi.org/10.3390/jpm13081268.
Moore, J., and Y. D. Guichot. 2024. “How to harness the power of health data to improve patient outcomes.” World Economic Forum Annual Meeting. World Economic Forum. January 5, 2024. https://www.weforum.org/stories/2024/01/how-to-harness-health-data-to-improve-patient-outcomes-wef24/.
Murphy, P. K., M. E. Sellers, S. H. Bonds, and S. Scott. 2024. The SEER Program’s longstanding commitment to making cancer resources available. JNCI Monographs 2024(65):118–122. https://doi.org/10.1093/jncimonographs/lgae028.
Myhre, P. L., J. Tromp, W. Ouwerkerk, D. S. W. Ting, K. F. Docherty, C. M. Gibson, and C. S. P. Lam. 2024. Digital tools in heart failure: Addressing unmet needs. The Lancet Digital Health 6(10):e755–e766. https://doi.org/10.1016/s2589-7500(24)00158-4.
NAM (National Academy of Medicine). n.d. Learning health system shared commitments. Available at: https://nam.edu/our-work/programs/leadership-consortium/learning-health-system/ (accessed January 14, 2026).
NAM. 2025a. An Artificial Intelligence Code of Conduct for Health and Medicine: Essential Guidance for Aligned Action. Washington, DC: The National Academies Press.https://doi.org/https://doi.org/10.17226/29087.
NAM. 2025b. Leadership consortium. Available at: https://nam.edu/our-work/programs/leadership-consortium/ (accessed December 26, 2025).
NAM. 2025c. Learning health system shared commitments. Available at: https://nam.edu/our-work/programs/leadership-consortium/learning-health-system/ (accessed January 6, 2026).
NAM. 2018. Procuring Interoperability: Achieving High-Quality, Connected, and Person-Centered Care. Washington, DC: The National Academies Press. https://doi.org/10.17226/27114.
NCQA (National Committee for Quality Assurance). n.d. HEDIS digital quality measures. Available at: https://www.ncqa.org/hedis/the-future-of-hedis/digital-measures/ (accessed January 4, 2026).
NICHD (Eunice Kennedy Shriver National Institute of Child Health and Human Development). 2022. Privacy Preserving Record Linkage (PPRL) for Pediatric COVID-19 Studies. Available at: https://www.nichd.nih.gov/sites/default/files/inline-files/NICHD_ODSS_PPRL_for_Pediatric_COVID-19_Studies_Public_Final_Report_508.pdf (accessed January 5, 2025).
NICHD. “Item of interest: NICHD-developed guide sets framework to link maternal, infant health data.” Press Release. June 26, 2023. Available at: https://www.nichd.nih.gov/newsroom/news/062623-maternal-guide (accessed January 4, 2026).
NIH (National Institutes of Health). 2025. Surveillance, epidemiology, and end results program. Available at: https://seer.cancer.gov/ (accessed December 29, 2025).
NQF (National Quality Forum). 2015. NQF-Endorsed Measures for Endocrine Conditions Final Report.
Nguyen, Q. C., E. M. Aparicio, M. Jasczynski, A. Channell Doig, X. Yue, H. Mane, N. Srikanth, F. X. M. Gutierrez, N. Delcid, X. He, and J. Boyd-Graber. 2024. Rosie, a health education question-and-answer chatbot for new mothers: Randomized pilot study. JMIR Formative Research 8:e51361. https://doi.org/10.2196/51361.
NCDIT (North Carolina Department of Information Technology). 2025. What is the digital divide? Available at: https://www.ncbroadband.gov/digital-divide/what-digital-divide (accessed December 29, 2025).
ONC, CDC, RCE (Office of the National Coordinator for Health Information Technology, Centers for Disease Control and Prevention, and the ONC TEFCA Recognized Coordinating Entity). 2023. Trusted exchange framework and common agreement update for the public health community. Available at: https://rce.sequoiaproject.org/wp-content/uploads/2023/01/TEFCA-PH-Exchange-SOP-CDC-ONC-RCE-Meeting-1.12.23_FINAL-v2.pdf (accessed December 26, 2025).
ONC HHS (Office of the National Coordinator for Health Information Technology, Department of Health and Human Services). 2020. “21st Century Cures Act: Interoperability, Information Blocking, and the ONC Health IT Certification Program.” Federal Register, 85 FR 25642, May 1, 2020. Available at: https://www.federalregister.gov/documents/2020/05/01/2020-07419/21st-century-cures-act-interoperability-information-blocking-and-the-onc-health-it-certification (accessed December 31, 2025).
Opiyo, N., C. Young, J. Harris Requejo, J. Erdman, S. Bales, and A. P. Betrán. 2020. Reducing unnecessary caesarean sections: Scoping review of financial and regulatory interventions. Reproductive Health 17:133. https://doi.org/10.1186/s12978-020-00983-y.
Orozco-Beltrán, D., S. Artola-Menéndez, A. Hormigo-Pozo, D. Cararach-Salami, J. L. Alonso-Jerez, E. Álvaro-Grande, C. Villabrille-Arias, F. J. de Toro-Santos, M. J. Galindo-Puerto, I. Marín-Jiménez, A. Gómez-García, R. Ledesma-Rodriguez, G. Fernández, and K. Ferreira de Campos. 2021. Healthcare experience among patients with type 2 diabetes: A cross-sectional survey using the IEXPAC tool. Endocrinology, Diabetes & Metabolism 4(2):e00220. https://doi.org/https://doi.org/10.1002/edm2.220.
Page II, R. L., C. L. O’Bryant, D. Cheng, T. J. Dow, B. Ky, C. M. Stein, A .P. Spencer, R. J. Trupp, and J. Lindenfeld, on behalf of the American Heart Association Clinical Pharmacology and Heart Failture and Transplantation Committees of the Council on Clinical Cardiology; Council on Cardiovascular Surgery and Anesthesia; Council on Cardiovascular and Stroke Nursing; and Council on Quality of Care and Outcomes Research. 2016. Drugs that may cause or exacerbate heart failure: A scientific statement from the American Heart Association. Circulation 134(6):e32–e69. https://doi.org/10.1161/CIR.0000000000000426.
Paul, M. K. 2024. Maternal navigation: For the common good. American Journal of Managed Care 30(Spec No. 10):sp745–sp750. https://doi.org/10.37765/ajmc.2024.89608.
PCI Security Standards Council. n.d. Home. Available at: https://www.pcisecuritystandards.org/ (accessed January 5, 2026).
Phelan, D., D. Gottlieb, J. C. Mandel, V. Ignatov, J. Jones, B. Marquard, A. Ellis, and K. D. Mandl. 2024. Beyond compliance with the 21st Century Cures Act Rule: A patient controlled electronic health information export application programming interface. Journal of the American Medical Informatics Association 31(4):901–909. https://doi.org/10.1093/jamia/ocae013.
Phibbs, C., S. Leonard, K. Kozhimannil, E. Main, C. Phibbs, S. Schmitt, and S. Lorch. 2020. Expanding the estimate of the costs of severe maternal morbidity to include readmissions, physician fees, and infant costs. Health Services Research 55(S1):143. https://doi.org/10.1111/1475-6773.13536.
Phibbs, C. M., K. B. Kozhimannil, S. A. Leonard, S. A. Lorch, E. K. Main, S. K. Schmitt, and C. S. Phibbs. 2022. The effect of severe maternal morbidity on infant costs and lengths of stay. Journal of Perinatology 42(5):611–616. https://doi.org/10.1038/s41372-022-01343-3.
Pinto, N. M., R. Nelson, M. Puchalski, T. D. Metz, and K. J. Smith. 2014. Cost-effectiveness of prenatal screening strategies for congenital heart disease. Ultrasound in Obstetrics & Gynecology 44(1):50–57. https://doi.org/10.1002/uog.13287.
Poissant, L., J. Pereira, R. Tamblyn, and Y. Kawasumi. 2005. The impact of electronic health records on time efficiency of physicians and nurses: A systematic review. Journal of the American Medical Informatics Association 12(5):505–516. https://doi.org/10.1197/jamia.M1700.
Potter, B. 2022. “How building codes work in the US.” Construction Physics. July 29, 2022. Available at: https://www.construction-physics.com/p/how-building-codes-work-in-the-us (accessed January 5, 2026).
Qiu, M., L.-L. Ding, M. Zhang, J.-H. Lin, X.-B. Wei, and H. Huang. 2020. GLP-1RAs and SGLT2is reduce cardiovascular events independent of reductions of systolic blood pressure and body weight: A meta-analysis with meta-regression. Diabetes Therapy 11(10):2429–2440. https://doi.org/10.1007/s13300-020-00912-z.
Rajesh, A. E., O. Q. Davidson, C. S. Lee, and A. Y. Lee. 2023. Artificial intelligence and diabetic retinopathy: AI framework, prospective studies, head-to-head validation, and cost-effectiveness. Diabetes Care 46(10):1728–1739. https://doi.org/10.2337/dci23-0032.
Reddy, V. Y., Y. Zhong, A. M. McGovern, S. L. Amorosi, M. B. Gavaghan, D. S. Hertz, K. Low, S. Freeman, and D. R. Holmes, Jr. 2024. Comparative costs to Medicare and Medicare beneficiaries of alternative AF stroke risk reduction strategies. Clinicoeconomics and Outcomes Research 16:81–96. https://doi.org/10.2147/ceor.S440556.
Rezende, P. C., M. A. Hlatky, W. Hueb, R. M. Rahmi Garcia, L. da Silva Selistre, E. G. Lima, C. L. Garzillo, T. L. Scudeler, G. A. B. Boros, F. F. Ribas, C. V. Serrano, Jr., J. A. F. Ramires, and R. Kalil Filho. 2020. Association of longitudinal values of glycated hemoglobin with cardiovascular events in patients with Type 2 diabetes and multivessel coronary artery disease. JAMA Network Open 3(1):e1919666. https://doi.org/10.1001/jamanetworkopen.2019.19666.
Rivera Rivera, J. N., K. E. AuBuchon, M. Smith, C. Starling, K. G. Ganacias, A. Danielson, L. Patchen, J. A. Rethy, H. J. Blumenthal, A. D. Thomas, and H. Arem. 2024. Development and refinement of a chatbot for birthing individuals and newborn caregivers: Mixed methods study. JMIR Pediatrics and Parenting 7:e56807. https://doi.org/10.2196/56807.
Sadik, H., D. Pritchard, D.-M. Keeling, F. Policht, P. Riccelli, G. Stone, K. Finkel, J. Schreier, and S. Munksted. 2022. Impact of clinical practice gaps on the implementation of personalized medicine in advanced non–small-cell lung cancer. JCO Precision Oncology 6:e2200246. https://doi.org/10.1200/po.22.00246.
Samal, L., P. C. Dykes, J. O. Greenberg, O. Hasan, A. K. Venkatesh, L. A. Volk, and D. W. Bates. 2016. Care coordination gaps due to lack of interoperability in the United States: A qualitative study and literature review. BMC Health Services Research 16:143. https://doi.org/10.1186/s12913-016-1373-y.
Santo, K., and J. Redfern. 2020. Digital health innovations to improve cardiovascular disease care. Current Atherosclerosis Reports 22(12):71. https://doi.org/10.1007/s11883-020-00889-x.
Sapna, F., F. Raveena, M. Chandio, K. Bai, M. Sayyar, G. Varrassi, M. Khatri, S. Kumar, and T. Mohamad. 2023. Advancements in heart failure management: A comprehensive narrative review of emerging therapies. Cureus 15(10):e46486. https://doi.org/10.7759/cureus.46486.
Scott, J. A., J. Lennerz, M. L. Johnson, L. N. Gordan, R. H. Dumanois, L. Quagliata, L. L. Ritterhouse, F. Cappuzzo, B. Wang, M. Xue, A. Vasudevan, P. Varughese, V. Vaidya, M. Gart, N. Dorrow, H. J. Gierman, and R. J. Choksi. 2024. Compromised outcomes in Stage IV non–small-cell lung cancer with actionable mutations initially treated without tyrosine kinase inhibitors: A retrospective analysis of real-world data. JCO Oncology Practice 20:145–153. https://doi.org/10.1200/op.22.00611.
SEC (U.S. Securities and Exchange Commission). n.d. About. Available at: https://www.sec.gov/about (accessed January 5, 2026).
Shaikh, A. R., I. P. Das, C. A. Vinson, and B. Spring. 2011. Cyberinfrastructure for consumer health. American Journal of Preventive Medicine 40(5)S91-S96. https://doi.org/10.1016/j.amepre.2011.02.012.
Shiffman, R. N., G. Michel, A. Essaihi, and E. Thornquist. 2004. Bridging the guideline implementation gap: A systematic, document-centered approach to guideline implementation. Journal of the American Medical Informatics Association 11(5):418–426. https://doi.org/10.1197/jamia.M1444.
Signe, L. 2023. Fixing the global digital divide and digital access gap. TechTank, the Brookings Institution. July 5, 2023. Available at: https://www.brookings.edu/articles/fixing-the-global-digital-divide-and-digital-access-gap/ (accessed January 4, 2026).
Sinsky, C. A., A. Rule, G. Cohen, B. G. Arndt, T. D. Shanafelt, C. D. Sharp, S. L. Baxter, M. Tai-Seale, S. Yan, Y. Chen, J. Adler-Milstein, and M. Hribar. 2020. Metrics for assessing physician activity using electronic health record log data. Journal of the American Medical Informatics Association 27(4):639–643. https://doi.org/10.1093/jamia/ocz223.
Sonawane, K., A. Garg, B. A. Toll, A. A. Deshmukh, and G. A. Silvestri. 2024. Lung cancer screening communication in the US, 2022. JAMA Network Open 7(11): e2442811. https://doi.org/10.1001/jamanetworkopen.2024.42811.
Starlink. n.d. Reliable high-speed internet from space. Available at: https://www.starlink.com/us (accessed January 5, 2026).
Stegman, M. M., E. Lucarelli-Baldwin, and S. H. Ural. 2023. Disparities in high risk prenatal care adherence along racial and ethnic lines. Frontiers in Global Womens Health 4:1151362. https://doi.org/10.3389/fgwh.2023.1151362.
Stokel-Walker, C. 2024. How patients are using AI. BMJ 387:q2393. https://doi.org/10.1136/bmj.q2393.
Sun, L., M. Hsu, R. B. Cohen, C. J. Langer, R. Mamtani, and C. Aggarwal. 2021. Association between KRAS variant status and outcomes with first-line immune checkpoint inhibitor-based therapy in patients with advanced non-small-cell lung cancer. JAMA Oncology 7(6):937–939. https://doi.org/10.1001/jamaoncol.2021.0546.
Sweet, C. C., C. B. Jasik, A. Diebold, A. DuPuis, and B. Jendretzke. 2020. Cost savings and reduced health care utilization associated with participation in a digital diabetes prevention program in an adult workforce population. Journal of Health Economics and Outcomes Research 7(2):139–147. https://doi.org/10.36469/jheor.2020.14529.
The Catalyze Tech Working Group. 2021. The ACT Report: Action to Catalyze Tech: A Paradigm Shift for DEI. Available at: https://www.actreport.com/download/ (accessed January 5, 2026).
Thomas, M. C., M. Brownlee, K. Susztak, K. Sharma, K. A. M. Jandeleit-Dahm, S. Zoungas, P. Rossing, P.-H. Groop, and M. E. Cooper. 2015. Diabetic kidney disease. Nature Reviews Disease Primers 1:15018. https://doi.org/10.1038/nrdp.2015.18.
Tsamantioti, E., A. Sandström, C. Lindblad Wollmann, J. M. Snowden, and N. Razaz. 2025. Association of severe maternal morbidity with subsequent birth. JAMA 333(2):133–142. https://doi.org/10.1001/jama.2024.20957.
Tsiachristas, A., K. Chan, E. Wahome, B. Kearns, P. Patel, M. Lyasheva, N. Syed, S. Fry, T. Halborg, H. West, E. Nicol, D. Adlam, B. Modi, A. Kardos, J. P. Greenwood, N. Sabharwal, G. L. De Maria, S. Munir, E. McAlindon, Y. Sohan, P. Tomlins, M. Siddique, C. Shirodaria, R. Blankstein, M. Desai, S. Neubauer, K. M. Channon, J. Deanfield, R. Akehurst, C. Antoniades, and the ORFAN Consortium. 2024. Cost-effectiveness of a novel AI technology to quantify coronary inflammation and cardiovascular risk in patients undergoing routine coronary computed tomography angiography. European Heart Journal – Quality of Care & Clinical Outcomes 11(4):434–444. https://doi.org/10.1093/ehjqcco/qcae085.
Tso, G. J., S. W. Tu, C. Oshiro, S. Martins, M. Ashcraft, K. W. Yuen, D. Wang, A. Robinson, P. A. Heidenreich, and M. K. Goldstein. 2017. Automating guidelines for clinical decision support: Knowledge engineering and implementation. AMIA Annual Symposium Proceedings Archive 2016:1189–1198. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC5333329/ (accessed January 4, 2026).
Valencia, Z., D. Bozzi, A. Sen, and K. Martin. 2022. The price of childbirth in the U.S. tops $13,000 in 2020. Health Care Cost Institute. May 10, 2022. Available at: https://healthcostinstitute.org/hcci-originals-dropdown/all-hcci-reports/the-price-of-childbirth-in-the-u-s-tops-13-000-in-2020 (accessed January 5, 2026).
Valishery, L. S. 2024. Cisco – Statistics & facts. Statista. December 17, 2025. Available at: https://www.statista.com/topics/2286/cisco/#topicOverview (accessed January 5, 2026).
Van den Berg, V. J., M. Strachinaru, K. M. Akkerhuis, S. Baart, M. Brankovic, A. A. Constantinescu, J. H. Cornel, O. C. Manintveld, V. A. W. M. Umans, D. Rizopoulos, M. L. Geleijnse, E. Boersma, B. M. van Dalen, and I. Kardys. 2019. Repeated echocardiograms do not provide incremental prognostic value to single echocardiographic assessment in minimally symptomatic patients with chronic heart failure: Results of the Bio-SHIFT study. Journal of the American Society of Echocardiography 32(8):1000–1009. https://doi.org/10.1016/j.echo.2019.04.419.
Venkatesh, K. K., R. A. Strauss, C. A. Grotegut, R. P. Heine, N. C. Chescheir, J. S. A. Stringer, D. M. Stamilio, K. M. Menard, and J. E. Jelovsek. 2020. Machine learning and statistical models to predict postpartum hemorrhage. Obstetrics & Gynecology 135(4):935–944. https://doi.org/10.1097/aog.0000000000003759.
Voigt, J., M. Sasha John, A. Taylor, M. Krucoff, M. R. Reynolds, and C. Michael Gibson. 2014. A reevaluation of the costs of heart failure and its implications for allocation of health resources in the United States. Clinical Cardiology 37(5):312–321. https://doi.org/10.1002/clc.22260.
Vook, F. W., A. Ghosh, E. Diarte, and M. Murphy. 2018. 5G new radio: Overview and performance. 2018 52nd Asilomar Conference on Signals, Systems, and Computers. 1247–1251. https://doi.org/10.1109/ACSSC.2018.8645228.
Vyas, D. A., D. S. Jones, A. R. Meadows, K. Diouf, N. M. Nour, and J. Schantz-Dunn. 2019. Challenging the use of race in the vaginal birth after cesarean section calculator. Women’s Health Issues 29(3):201–204. https://doi.org/10.1016/j.whi.2019.04.007.
Warner Stevenson, L., H. J. Ross, L. D. Rathman, and J. P. Boehmer. 2023. Remote monitoring for heart failure management at home: JACC scientific statement. Journal of the American College of Cardiology 81(23):2272–2291. https://doi.org/10.1016/j.jacc.2023.04.010.
Westcott, J. M., F. Hughes, W. Liu, M. Grivainis, I. Hoskins, and D. Fenyo. 2022. Prediction of maternal hemorrhage using machine learning: Retrospective cohort study. Journal of Medical Internet Research 24(7):e34108. https://doi.org/10.2196/34108.
Wienert, J., T. Jahnel, and L. Maaß. 2022. What are digital public health interventions? First steps toward a definition and an intervention classification framework. Journal of Medical Internet Research 24(6):e31921. https://doi.org/10.2196/31921.
World Economic Forum. 2019. “4 ways data is improving healthcare.” Global Innovation Index. December 5, 2019. https://www.weforum.org/stories/2019/12/four-ways-data-is-improving-healthcare/.
World Wide Web Consortium. 2025. About us. Available at: https://www.w3.org/about/ (accessed December 27, 2025).
Wu, A. W. 2024. Chatting together: Using AI chatbots to improve diagnostic excellence. Journal of Patient Safety and Risk Management 29(5):222–224. https://doi.org/10.1177/25160435241292868.
Zeitlin, J., N. N. Egorova, T. Janevic, P. L. Hebert, E. Lebreton, A. Balbierz, and E. A. Howell. 2019. The impact of severe maternal morbidity on very preterm infant outcomes. The Journal of Pediatrics 215:56–63. https://doi.org/10.1016/j.jpeds.2019.07.061.
Zhao, X., S. Wu, B. Yan, and B. Liu. 2024. New evidence on the real role of digital economy in influencing public health efficiency. Scientific Reports 14:7190. https://doi.org/10.1038/s41598-024-57788-3.
Zhou, K., L. Donnelly, J. Yang, M. Li, H. Deshmukh, N. Van Zuydam, E. Ahlqvist, The Wellcome Trust Case Control Consortium, C. C. Spencer, L. Groop, A. D. Morris, H. M. Colhoun, P. C. Sham, M. I. McCarthy, C. N. A. Palmer, and E. R. Pearson. 2014. Heritability of variation in glycaemic response to metformin: A genome-wide complex trait analysis. The Lancet Diabetes & Endocrinology 2(6):481–487. https://doi.org/10.1016/s2213-8587(14)70050-6.

